The
cytochrome P450 (CYP) is a well known superfamily of enzymes that are
responsible for the oxidative and reductive metabolic transformation of
medications used in clinical practice.1-3 In addition, the CYP enzymes
are commonly associated with causing many clinically relevant drug-drug
interactions. Of the CYP enzymes, CYP3A4 is not only the most prevalent
CYP enzyme in the liver, but is used by more than 50% of medications on the
market for their metabolism and elimination from the body.1 In addition,
the CYP3A4 activity can be induced (or accelerated) or it can be inhibited
(decreased), thereby changing the drug concentrations present in the body and
its pharmacokinetic profile.1-4 The inhibition of CYP3A4 can result in the accumulation of parent drug
concentrations that can put the patient at increased risk for side effects and
possible toxicity.
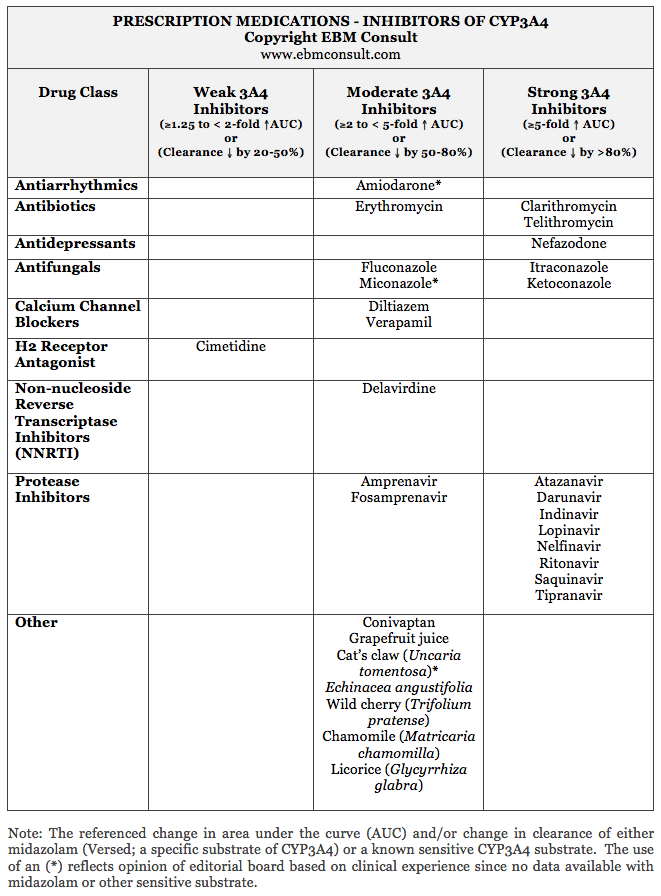
While the below table is clearly not an exhaustive list of every medication
known to inhibit CYP3A4, these are the most common medications used in clinical
practice that are known to interact with other medications that are substrates
of CYP3A4. It is also important to note that not all medications within a
particular drug class have the same effect. For example, within the
macrolide antibiotics, all of them are known inhibitors of CYP3A4 with the exception
of azithromycin. For the calcium channel blockers, it is only the
non-dihydropyridine calcium channel blockers that are known inhibitors of
CYP3A4, but not amlodipine or nifedipine. Lastly, within the
non-nucleoside reverse transcriptase inhibitors (NNRTI) used in the management
of HIV, only delavirdine is an inhibitor of CYP3A4 whereas the other NNRTIs in
the class are considered to be inducers of CYP3A4.7-9 This is important
as it reveals that the pharmacokinetic profiles do not always completely follow
a class effect. Therefore, anytime the medications listed in the provided table
are initiated in a patient already on stable does of other medications, the
chances of a clinically relevant drug interaction is likely and should be taken
into consideration upon initiation. The medications known to be CYP3A4
inhibitors are summarized in the below table based on their class of
medications and classifications.1,2,5,6
References:
- Rendic S, Ci Carlo FJ. Human cytochrome P450 enzymes: a status
report summarizing their reactions, substrates, inducers, and
inhibitors. Drug Metab Rev 1997;29:413-580.
- United
States Food and Drug Administration. Guidance for Industry. Drug
Interaction Studies - Study Design, Data Analysis, and Implications for
Dosing and Labeling. September 2006. Clinical Pharmacology. Accessed
last on 5/19/2009.
- Ohno
Y, Hisaka A, Suzuki H. General framework for the quantitative
prediction of CYP3A4-mediated oral drug interactions based on the AUC
increase by coadministration of standard drugs. Clin Pharmacokinet
2007;46:681-96.
- Busti
AJ, Herrington JD, Daves BJ, McKeever GC. What is the process to
determine if medications are to be classified as weak, moderate or
strong inhibitors of CYP3A4? PW Drug Interact Newsl 2009;1(39):1-3
- Delavirdine (Rescriptor®) product package insert. Pfizer Inc. New York, NY. May 2008.
- Conivaptan (Vaprisol®) product package insert. Astellas Pharma US, Inc. Deerfield, IL. October 2008.
- Efavirenz (Sustiva) product package insert. Bristol-Myers Squibb. Princeton, NJ. March 2008. Last accessed 1-22-2009.
- Nevirapine (Viramune) product package insert. Boehringer Ingelheim Pharmaceuticals. Last accessed on 1-22-2009.
- Gerber
JG, Rosenkranz SL, Fichtenbaum CJ et al. Effect of efavirenz on the
pharmacokinetics of simvastatin, atorvastatin, and pravastatin: results
of AIDS Clinical Trials Group 5108 Study. J Acquir Immune Defic Syndr
2005;39:307-12.






