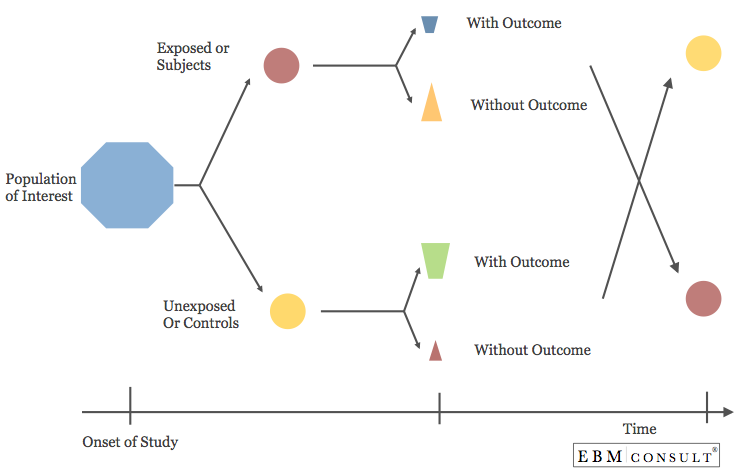
Cross-Over Study Design
|
|---|
- A study design where all patients from a population of interest are initially assigned to one of two groups, one group who gets exposed to the intervention or a second group that does not get exposed to the intervention.
- After a period of time, an evaluation of the outcome is done
and the patients from both groups undergo a period of washout so that the
effect from the initial group intervention has been removed. Once this occurs,
the subjects will then cross-over into the other group where the process starts
over.
- This study design provides the advantage of reducing the variability in outcome measures from outside confounders because each patient will serve as his or her own control.
- Reduced influence by confounders since patients serve as their own controls
- Reduced variability in the outcome(s) being measured, thus increasing the precision of estimation
- Smaller sample sizes required
- Having the opportunity to receive both treatments can sometimes be attractive for subjects
- Cannot be done when the subjects can only receive one treatment
- Assumption of no carryover effects (from washout period) is difficult to sometimes accurately test
- May take longer than a randomized clinical trial since patients have to cross over into each arm after an appropriate washout period
- Can be subject to period effects where differences in the effectiveness of an intervention can occur due to the passage of time. For example:
- Development of tolerance
- Resistance
- Dropouts
- Changes in the disease process being evaluated or treated
Description of a Cross-Over Study
Advantages
Disadvantages