Nitrofurantoin (Macrobid, Macrodantin): Drug Monograph
|
|---|
- Urinary tract infections due to susceptible strains of Escherichia coli, enterococci, Staphylococcus aureus, and certain susceptible strains of Klebsiella andEnterobacter species
- Note: Not indicated for the treatment of pyelonephritis or perinephric abscesse
- General Notes: Should be taken with food
- (Macrobid) Acute or Recurrent Uncomplicated UTI:
- 100 mg twice daily for 7 days
- Surgical prophylaxis - 100 mg twice daily on the day of the procedure and 3 days thereafter
- (Macrodantin) UTI:
- 50-100 mg four times a day for 1 week or for at least 3 days after sterility of the urine obtained
- Long-term suppressive therapy: Reduce dose to 50-100 mg at bedtime
- Macrobid, UTIs, >12 years:
- 100 mg every 12 hours for 7 days
- Macrodantin, UTIs, ≥1 month of age:
- 5-7 mg/kg/day given in 4 divided doses for 1 week or for at least 3 days after sterility of the urine obtained
- Long-term suppressive therapy: 1 mg/kg/day given in a single dose or in 2 divided doses
- CrCl <60 mL/min or clinically significant elevated serum creatinine are contraindications
- Impaired renal function (CrCl < 60 mL/min or clinically elevated serum creatinine) - increased risk of toxicity (e.g., pulmonary fibrosis with long-term use) because of impaired excretion of the drug
- Pregnant patients at term (38-42 weeks gestation), during labor and delivery, or when the onset of labor is imminent - because of the possibility of hemolytic anemia due to immature erythrocyte enzyme systems (glutathione instability) or underlying presence of G6PD deficiency
- Previous history of cholestatic jaundice/hepatic dysfunction associated with nitrofurantoin
- Known hypersensitivity to nitrofurantoin
- Pulmonary reactions - acute, subacute, or chronic pulmonary reactions possible. Discontinue drug if reactions occur. Pulmonary reactions can be a contributing cause of death. Chronic pulmonary reactions (diffuse interstitial pneumonitis or pulmonary fibrosis, or both) can develop insidiously. Reactions are rare and generally occur in patients receiving therapy for six months or longer. Close monitoring of the pulmonary condition of patients receiving long-term therapy is warranted.
- Hepatotoxicity - reactions may include hepatitis, cholestatic jaundice, chronic active hepatitis, and hepatic necrosis. Monitor patients periodically for changes in biochemical tests that would indicate liver injury. Withdraw drug immediately if hepatitis occurs.
- Neuropathy - conditions such as renal impairment, anemia, diabetes mellitus, electrolyte imbalance, vitamin B deficiency, and debilitating disease may enhance occurrence. Monitors patients receiving long-term therapy.
- Hemolytic anemia - hemolysis appears to be linked to a glucose-6-phosphate dehydrogenase deficiency in the red blood cells of the affected patients. Discontinue drug if hemolysis occurs.
- Clostridium difficile-associated diarrhea (CDAD) - may range in severity from mild diarrhea to fatal colitis. Ongoing antibiotic use not directed against C. difficile may need to be discontinued.
- Nausea
- Headache
- Flatulence
- Diarrhea
- Dizziness, drowsiness, amblyopia
- Acute pulmonary hypersensitivity reaction
- Pruritus, urticaria
- Alopecia
- Fever, chills, malaise
- Superinfection caused by resistant organisms
- Symptoms may include vomiting.
- Induction of emesis is recommended.
- A high fluid intake should be maintained to promote urinary excretion of the drug. Nitrofurantoin is dialyzable.
- Antacids containing magnesium trisilicate - reduce both the rate and extent of absorption of nitrofurantoin.
- Uricosuric drugs (probenecid and sulfinpyrazone) - can inhibit renal tubular secretion of nitrofurantoin. The resulting increase in serum levels may increase toxicity, and the decreased urinary levels could lessen its efficacy as a urinary tract antibacterial.
- Pregnancy: Pregnancy Category B (except should be avoided after 38 weeks)
- Labor and Delivery: Contraindicated during labor and delivery.
- Nursing Mothers: Nitrofurantoin has been detected in human breast milk in trace amounts. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
- Renal Impairment: Impaired renal function (CrCl <60 mL/min or clinically significant elevated serum creatinine) are contraindications.
- Hepatic Impairment: None
- Pediatric Patients: Contraindicated in infants below the age of one month. Safety and effectiveness in patients below the age of twelve years have not been established.
- Geriatric Patients: Spontaneous reports suggest a higher proportion of pulmonary reactions in the elderly, including fatalities; these differences appear to be related to the higher proportion of elderly patients receiving long-term nitrofurantoin therapy. In general, the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease other other drug therapy in elderly patients should be considered when prescribing nitrofurantoin. Monitor renal function.
- Nitrofurantoin has been detected in human breast milk in trace amounts. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
-
Scientific Name:
-
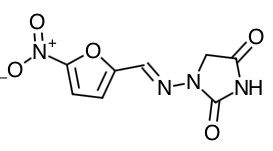
Nitrofurantoin macrocrystals: 1-[[[5-nitro-2-furanyl]methylene] amino]-2,4-imidazolidinedione
-
Nitrofurantoin monohydrate: 1-[[[5-nitro-2-furanyl]methylene] amino]-2,4- imidazolidinedione monohydrate.
-
Empirical Formula: C8H6N4O5
-
Molecular Weight: Nitrofurantoin macrocrystals: 238.16 Nitrofurantoin monohydrate: 256.17
- The exact mechanism is unknown but appears to be related to formation of reactive intermediates by bacterial flavoproteins. The reactive intermediates are thought to damage and inhibit cellular pathways including protein synthesis, cell wall synthesis and DNA/RNA synthesis.
- Absorption: Bioavailability is about 40% when taken with food
- Distribution: Found in breast milk
- Metabolism: None
- Elimination: Macrobid: Urine (20-25% unchanged)
- Macrodantin: Urine (100 mg four times a day for 7 days: Day 1: 37.9%; Day 7: 35%)
- Patients should be advised to take nitrofurantoin with food (ideally breakfast and dinner) to further enhance tolerance and improve drug absorption.
- Patients should be instructed to complete the full course of therapy; however, they should be advised to contact their physician if any unusual symptoms occur during therapy.
- Patients should be advised not to use antacid preparations containing magnesium trisilicate while taking nitrofurantoin.
- Patients should be advised that antibacterial drugs including nitrofurantoin should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). The medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may decrease the effectiveness of the immediate treatment and increase the likelihood that bacteria will develop resistance and will not be treatable by nitrofurantoin or other antibacterial drugs in the future.
- Patients should be instructed that diarrhea is a common problem caused by antibiotics, which usually ends when the antibiotic is discontinued. Patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
- Pharmacology: The Mechanism and Cross-Reactivity in Penicillin and Cephalosporin Antibiotics
- Drug Monograph: Aztreonam (Azactam; Cayston)
- Drug Monograph: Ceftriaxone (Rocephin)
- Drug Monograph: Linezolid (Zyvox)
Indications
Dosing
(Adult)
(Pediatrics)
Renal Dosing
Contraindications
Warnings
Adverse Reactions
Overdose
Drug Interactions
Special Populations
Breastfeeding
Chemical Structure
Mechanism of Action
Pharmacokinetics
Counseling Points
Other EBM Consult Related Content
MESH Terms & Keywords
|
|---|
|





