Felodipine (Plendil): Drug Monograph
|
|---|
- Treatment of mild to moderate essential hypertension.
- May be used alone or concomitantly with other antihypertensive agents.
- General Notes:
- The consumption of grapefruit juice prior to or during treatment should be avoided due to pharmacodynamic effect.
- Should be swallowed whole; not crushed or chewed.
- The tablets should not be taken with a meal rich in carbohydrates or fat.
- Hypertension:
- 5 mg by mouth once daily.
- May decrease to 2.5 mg every day or increase to 10 mg every day at intervals of not <2 weeks, depending on the patient's response.
- Elderly (over 65 years): 2.5 mg once daily
- No FDA approved dosing, but children > 6 years of age can be started on 2.5 mg once daily and titrated to a max dose of 10 mg once daily.
- Known hypersensitivity to felodipine
- Known hypersensitivity to other dihydropyridines
- In women of childbearing potential, in pregnancy, and during lactation
- Congestive heart failure - use caution, particularly in combination with a beta-blocker.
- Hypotension, myocardial ischemia- careful monitoring of blood pressure recommended
- Beta-blocker withdrawal
- Outflow obstruction - use with caution in the presence of left ventricular outflow obstruction
- Dermatologic lesion - may include leucocytoclastic vasculitis, rash and flush
- Peripheral edema
- Headache
- Asthenia
- Palpitation
- Warm sensation/flushing
- Nausea
- Dyspepsia
- Constipation
- Dizziness Paresthesia
- Upper respiratory infection
- Cough
- Rash
- Can cause excessive peripheral vasodilation with marked hypotension and possibly bradycardia
- Activated charcoal may be used
- Symptomatic treatment in case of severe hypotension
- When accompanied by bradycardia, 0.5-1 mg atropine should be administered intravenously
- Sympathomimetic drugs predominantly affecting the a1-adrenoceptor may be given if needed
- Hemodialysis removal has not been established
- Cytochrome P450 Enzyme substrates - includes benzodiazepines, flecainide, imipramine, propafenone, terfenadine, theophylline
- Cytochrome P450 enzyme inhibitors - includes azole antifungals (ketoconazole and itraconazole), cimetidine, cyclosporine, erythromycin, quinidine, warfarin - cause an increase in felodipine plasma concentrations
- CYP3A4 enzyme inducers - includes St. John's Wort, phenobarbital, phenytoin, rifampin - cause a decrease in plasma concentrations of felodipine
- Metoprolol -increases the AUC and Cmax of metoprolol
- Digoxin - when felodipine as conventional tablets is given, the Cmax of digoxin is significantly increased; with extended release formulation, no change in Cmax
- Tacrolimus - may increase the concentration of tacrolimus
- Pregnancy: Pregnancy Category C
- Labor and Delivery: None
- Nursing Mothers: It is not known whether felodipine is secreted in human milk. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
- Renal Impairment:
- Hepatic Impairment: May have elevated plasma concentrations of felodipine and therefore, may require lower dosage. Monitor blood pressure closely during initial administration and after dosage adjustment. A dosage of 10 mg daily should not be exceeded.
- Pediatric Patients: Safety and efficacy have not been established.
- Geriatric Patients: Patients over 65 years of age may have elevated plasma concentrations of felodipine and may require lower dosage. (A dosage of 10 mg daily should not be exceeded).
- It is not known whether felodipine is secreted in human milk. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
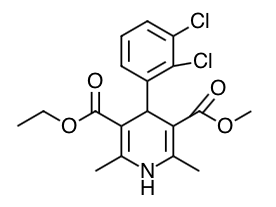
- Scientific Name: 3,5-pyridinedicarboxylic acid, 4-(2,3-dichlorophenyl)-1,4-dihydro 2,6-
dimethyl-, ethyl methyl ester
- Empirical Formula: C18H19Cl2NO4
- Molecular Weight: 384.26
- Selectively inhibits the transmembrane influx of calcium ions into cardiac muscle and vascular smooth muscle, thereby decreasing the contractile processes of these tissues.
- In vitro studies show that the effects of felodipine on contractile mechanisms are selective, with greater effects on vascular smooth muscle than on cardiac muscle. Negative inotropic effects can be detected in vitro, but such effects have not been seen in intact animals.
- The effect of felodipine on blood pressure in man is principally a consequence of a dose- related decrease in peripheral vascular resistance, with a modest reflex increase in heart rate.
- Reduces total peripheral resistance, which leads to a decrease in blood pressure associated with a modest reflex increase in heart rate. This reflex increase in heart rate frequently occurs during the first week of therapy and generally attenuates over time. Heart rate increases of 5-10 beats per minute may be seen during chronic administration.
- Following administration of felodipine a reduction in blood pressure generally occurs within two to five hours.
- During chronic administration, substantial blood pressure control lasts for approximately 24 hours; reductions in diastolic blood pressure at trough plasma levels were 40-60% of those at peak plasma levels.
- The antihypertensive effect is dose-dependent and correlates with the plasma concentration of felodipine.
- Felodipine in therapeutic doses has no effect on conduction in the conducting system of the heart and no effect on the A-V nodal refractoriness. No direct additional effects to those registered after beta-blockade are observed when PLENDIL is given concomitantly.
- Renal vascular resistance is decreased by felodipine while glomerular filtration rate remains unchanged. Mild diuresis, natriuresis and kaliuresis have been observed during the first week of therapy. No significant effects on serum electrolytes have been observed during short and long-term therapy. No general salt and water retention occurs during long-term therapy.
- Absorption: Due to rapid biotransformation of felodipine during its first pass through the portal circulation, the systemic availability is approximately 15%.
- The peak plasma concentration of felodipine (Cmax) is significantly increased by 1.5 to 2 fold when it is taken after a high fat or high carbohydrate meal versus fasting. Because the effects of felodipine on blood pressure are related to plasma levels, this increase in Cmax may cause a clinically significant fall in blood pressure
- Therefore felodipine should not be administered with meals rich in carbohydrate or fat
- Protein Binding: The plasma protein binding is approximately 99%. It is bound predominately to the albumin fraction.
- Half-Life: The mean terminal half-life was 24.5±7.0 hours and 27.5±8.4 hours in elderly hypertensive patients and 14.1±5.6 hours in young healthy volunteers.
- Distribution: Vd=10 L/kg
- Metabolism: Felodipine is extensively metabolized in the liver, predominantly by cytochrome P-450 CYP3A4.
- Elimination: After 72 hours, approximately 70% of a given dose is excreted as metabolites in the urine and 10% is secreted in the feces. Less than 0.5% of a dose is recovered unchanged in the urine. Six metabolites, which account for 23% of the oral dose, have been identified: none has significant vasodilating activity.
- Inform the patient to take the medication exactly as prescribed, and to not crush or chew the tablets.
- Instruct patients that mild gingival hyperplasia (gum swelling) may occur during treatment. Good dental hygiene decreases its incidence and severity.
- Instruct women of child bearing age that if they are pregnant or plan to become pregnant, to advise their doctor before initiating treatment. If she becomes pregnant while using felodipine, inform her doctor as soon as possible.
- Inform the patient to tell the doctor or pharmacist about any medicines they are taking or have recently taken, including over-the-counter and herbal medicines.
- Instruct patients not to drink grapefruit juice or eat grapefruit while on felodipine.
- Instruct patients not to drink alcohol while on treatment as it may cause dizziness or a drop in blood pressure.
- Instruct patients to keep their doctor informed of all side effects, especially if they experience any of the following for more than a week: swelling of the ankles, a racing heartbeat, flushing or a feeling of warmth, headache, dizziness, or unusual tiredness.
- Drug Monograph: Amlodipine (Norvasc)
- Drug Monograph: Nicardipine (Cardene)
- Drug Monograph: Nimodipine (Nymalize)
- Drug Monograph: Amiodarone (Cordarone; Pacerone)
- Anatomy: Heart (External Anterior & Posterior) Anatomy
Indications
Dosing
(Adult):
(Pediatrics):
Contraindications
Warnings
Adverse Reactions
Overdose
Drug Interactions
Special Populations
Breastfeeding
Chemical Structure
Mechanism of Action
Pharmacodynamics
Pharmacokinetics
Counseling Points
Other EBM Consult Related Content
MESH Terms & Keywords
|
|---|
|





