Niacin or nicotinic acid is an antilipemic
agent used to treat dyslipidemia. Its role as a second-line therapy in the
treatment of dyslipidemia was established by niacin's ability to significantly
raise high-density lipoprotein cholesterol (HDL-C), as well as lowering
low-density lipoprotein cholesterol (LDL-C), lipoprotein A, and triglycerides.1
However, its use in practice is often limited by its toxicities. Niacin has
been associated with flushing, gastrointestinal upset, and hepatotoxicity. All
can influence continuation of therapy and are closely related to a particular
niacin formulation.1,2,8 The immediate release formulation has been associated
with higher incidences of flushing while the over-the-counter sustained release
(SR) formulation is more commonly associated with a higher incidence of
gastrointestinal side effects and hepatotoxicity. In contrast, the extended
release formulation available by prescription has the least incidence of side
effects.8
Drug-induced
hepatotoxicity can be classified as either a direct hepatotoxic mechanism or an
immune-mediated mechanism that is caused by a drug or its metabolites. The
injury from the drug in question will then result in hepatitis, cirrhosis,
cholestasis, steatosis, sinusoidal damage, or a blending of these
presentations.3,4 In regard to niacin hepatotoxicity, it is a direct effect
that will result in acute hepatitis or a mixture of acute hepatitis and
cholestasis.5
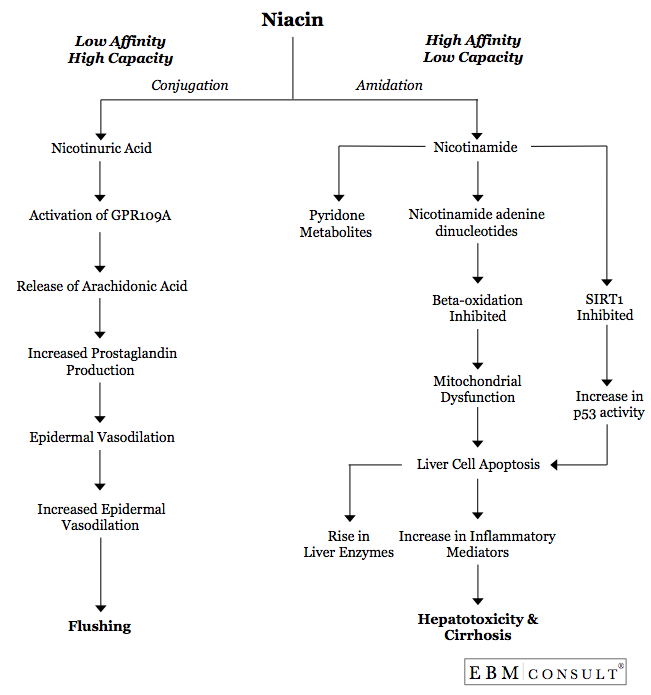
Once
niacin enters the liver, it is metabolized by either conjugation or amidation
(see Figure). The amidation pathway is associated with hepatotoxicity and is
classified as a high-affinity and low-capacity pathway. Thus, if niacin is
available in smaller concentrations over time, such as with a SR formulation,
the amidation pathway is unlikely to be saturated. Because it is a
high-affinity pathway, this will be the primary means for metabolism whenever
SR niacin is administered. When this pathway is primarily used, the
series of oxidative and reductive reactions result in the production of
nicotinamide adenine dinucleotide (NAD), which is associated with niacin's
hepatoxicity.6-8
The
NAD causes oxidative stress on the liver by inhibiting β-oxidation through the
electron transport chain leading to mitochondrial dysfunction. The
dysfunctional mitochondria fail to produce ATP in these cells leading to
apoptosis or necrosis.9 The necrosis of cells potentiates the release of IL-12,
IL-18, TNF-α, IFN-γ, and Fas from both Kupffer Cells and natural killer cells,
which initiates and propagates inflammatory responses and tissue damage. 3,4
Finally, there are data to suggest that nicotinamide itself may cause
propagation of apoptosis by inhibiting SIRT1, a regulator of p53 and inhibitor
of apoptosis.10 Overall, apoptosis and the resulting release of inflammatory
mediators leads to an increase in liver enzymes, tissue and sinusoidal damage,
and ultimately hepatotoxicity.3,4,9,10
References:
- Stone NJ et al. Circulation 2014;129:S1-45. and Grundy
SM, Becker D, Clark LT et al. Coordinating Committee of the National
Cholesterol Education Program. Implications of recent clinical trials for the
National Cholesterol Education Program Adult Treatment Panel III Guidelines.
Circulation 2004; 110: 227-39.
- Malloy
MJ and Kane JP. Chapter 35. Agents Used in Dyslipidemia. In: Katzung BG,
Masters SB, Trevor AJ, eds. Basic & Clinical Pharmacology. 12th ed. New
York: McGraw-Hill; 2012.
- Lee
WM. Drug-Induced Hepatotoxicity. N Engl J Med 2003;349:474-85.
- Holt
MP and Ju Cynthia. Mechanisms of Drug-Induced Liver Injury. AAPS J 2006;
8(1):E48-E54.
- Dalton
TA and Berry RS. Hepatotoxicity Associated with Sustained-Release Niacin. Am J
Med 1992;93:102-104.
- Dunatchik
AP, Ito MK, and Dujovne CA. A systematic review on evidence of the
effectiveness and safety of a wax-matrix niacin formulation. J Clin Lipidol
2012;6(2):121-31.
- Piepho
RW. The Pharmacokinetics and Pharmacodynamics of Agents Proven to Raise
High-Density Lipoprotein Cholesterol. Am J Cardiol 2000;86(suppl):35L-40L.
- Piper
JA. Overview of niacin formulations: Differences in pharmacokinetics, efficacy,
and safety. Am J Health Syst Pharm 2003;60 (suppl):S9-S14.
- Russman
S, Kullak-Ublick GA, and Grattagliano I. Current concepts of mechanisms in
drug-induced hepatotoxicity. Curr Med Chem 2009;16(23):3041-3053.
- Bitterman
KJ, Anderson RM, Cohen HY, Latorre-Esteves M, and Sinclair DA. Inhibition of
silencing and accelerated aging by nicotinamide, a putative negative regulator
yeast Sir1 and human SIRT1. J Biol Chem 2002;277:45099-45107.