Tobramycin (Bethkis, Tobi): Drug Monograph
|
|---|
- Inhalation Formulations (Only):
- Management of cystic fibrosis patients with Pseudomonas aeruginosa
- Safety and efficacy have not been demonstrated in patients under the age of six years, patients with a forced expiratory volume in less than one second (FEV1) less than 40% or greater than 80% predicted, or patients colonized with Burkholderia cepacia.
- Cystic Fibrosis (CF):
- Nebulized Solution (Bethkis; Kitabis Pak):
- 300 mg by inhalation every 12 hours x 28 days and then off for 28 days.
- Use only by oral inhalation.
- Administer nebulized solution by using a hand-held Pari LC Plus reusable nebulizer with a Pari Vios air compressor over approximately 15 minutes and until sputtering from the output of the nebulizer has occurred for at least 1 minutes.
- Do not mix with other medicines in the nebulizer. Administer other inhaled medicines (e.g., bronchodilators) before administration of therapy.
- Refer to PI for further preparation and administration instructions
-
Capsules (Tobi Podhaler):
- 112 mg by inhalation every 12 hours (given as 4 of the 28 mg capsules) x 28 days and then off for 28 days
- Do not swallow capsules.
- Use with Podhaler device only; always use the new Podhaler device provided with each weekly pack.
- Only remove capsule from blister immediately before use.
- ≥6 years: 300 mg twice daily by oral inhalation (as close to 12 hours apart as possible; not <6 hours apart) in repeated cycles of 28 days on drug, followed by 29 days off drug
- Capsules: Inhalation of four 28 mg capsules twice a day (as close to 12 hours apart as possible; not <6 hours apart) using the Podhaler device in repeated cycles of 28 days on drug, followed by 28 days off drug
- Solution, inhalation: 300 mg/4mL (Bethkis); 300 mg/5 mL (Tobi)
- Capsules, inhalation: 28 mg [8, 26, 224 capsules] (Tobi)
- Patients with known or suspected auditory, vestibular, renal or neuromuscular dysfunction - use caution when prescribing
- Aminoglycoside may aggravate muscle weakness because of a potential curare-like effect on neuromuscular function
- Bronchospasm - may occur with inhalation
- Audiograms, serum concentration, and renal function - should be monitored as appropriate
- Fetal harm - may occur when administered to a pregnant woman. Apprise women of the potential hazard to the fetus
- Forced expiratory volume decrease
- Rales
- Red blood cell sedimentation rate increase
- Dysphonia
- No overdoses have been reported in clinical trials.
- Signs and symptoms of acute toxicity from overdosage might include dizziness, tinnitus, vertigo, loss of high-tone hearing acuity, respiratory failure, and neuromuscular blockade.
- Administration by inhalation results in low systemic bioavailability of tobramycin.
- Not significantly absorbed following oral administration; serum concentrations may be helpful in monitoring overdosage.
- The possibility of drug interactions with alterations in drug disposition should be considered.
- Other drugs with neurotoxic or ototoxic potential - avoid concurrent and/or sequential use
- Ethacrynic acid, furosemide, urea, or mannitol - do not administer concomitantly
- Pregnancy: Pregnancy Category D
- Labor and Delivery: The safety and efficacy have not been studied in the puerperal patient.
- Nursing Mothers: Discontinue drug or nursing, taking into consideration the importance of the drug to a mother.
- Renal Impairment: The risk of adverse reactions to this drug may be greater in patients with impaired renal function. Patients with serum creatinine >2 mg/dL and blood urea nitrogen (BUN) >40 mg/dL have not been included in clinical studies and there are no data in this population to support a recommendation for or against dose adjustment.
- Hepatic Impairment: None
- Pediatric Patients: The safety and efficacy have not been studied in pediatric cystic fibrosis patients under six years of age.
- Geriatric Patients: The safety and efficacy have not been studied in adult cystic fibrosis patients over 31 years of age.
- Discontinue drug or nursing, taking into consideration the importance of the drug to a mother.
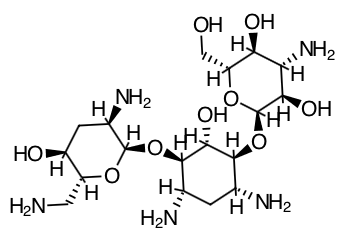
- Scientific Name: O-3-amino-3- deoxy-α-D-glucopyranosyl-(1→4)-O-[2,6-diamino- 2,3,6- trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine
- Empirical Formula: C18H37N5O9
- Molecular Weight: 467.52
- Tobramycin is an antibacterial drug. Acts primarily by disrupting protein synthesis in the bacterial cell which eventually leads to death of the cell.
- Tobramycin is a cationic polar molecule that does not readily cross epithelial membranes. The bioavailability of tobramycin may vary because of individual differences in nebulizer performance and airway pathology. Following administration, tobramycin remains concentrated primarily in the airways.
- Sputum Concentrations: Thirty minutes after inhalation of the first 300 mg dose of tobramycin, the maximum geometric mean concentration of tobramycin was 814 mcg/g (ranging from 23 to 2843 mcg/g) in sputum. High variability of tobramycin concentration in sputum was observed. Three hours after inhalation started, sputum tobramycin concentrations declined to approximately 15% of those observed at 30 minutes. After four weeks of therapy with tobramycin average mean sputum tobramycin concentrations obtained 10 minutes following administration were 717 mcg/g.
- Absorption: None
- Distribution: Crosses placenta
- Metabolism: None
- Elimination: The elimination half-life of tobramycin from serum is approximately two hours after intravenous (IV) administration. The elimination half-life following the inhalation of tobramycin is approximately 4.4 hours. Assuming tobramycin absorbed following inhalation behaves similarly to tobramycin following intravenous administration, systemically absorbed tobramycin is eliminated principally by glomerular filtration. Una bsorbed tobramycin following inhalation is likely eliminated in expectorated sputum.
- Patients should be informed that information on the long-term efficacy and safety of tobramycin is limited. There is no information in patients with severe cystic fibrosis (FEV1<40% predicted).
- Patients should be advised to complete a full 28-day course of tobramycin, even if they are feeling better. After 28 days of therapy, patients should stop treatment for the next 28 days, and then resume therapy for the next 28 day on and 28 day off cycle.
- Advise patients that are taking several different inhaled medications and/or performing chest physiotherapy the order they should take the therapies. It is recommended that tobramycin should be taken last.
- Tobramycin is to be used with the PARI LC PLUS reusable nebulizer and the PARI VIOS air compressor. Refer to the manufacturer's instructions for care and use of the nebulizer and compressor.
- Inform patients that ototoxicity, as measured by complaints of hearing loss or tinnitus, was reported by patients treated with tobramycin. Physicians should consider an audiogram at baseline, particularly for patients at increased risk of auditory dysfunction. If a patient reports tinnitus or hearing loss during therapy, the physician should refer that patient for audiological assessment. Patients should be reminded that vestibular toxicity may manifest as vertigo, ataxia, or dizziness.
- Inform patients that bronchospasm can occur with inhalation of tobramycin.
- Inform patients of adverse reactions associated with aminoglycosides such as nephrotoxicity and neuromuscular disorders.
- Inform patients of the need to monitor hearing, serum concentrations of tobramycin, or renal function as necessary during treatment.
- Inform patients that aminoglycosides can cause fetal harm when administered to a pregnant woman. Advise them to inform their doctor if they are pregnant, become pregnant, or plan to become pregnant.
- Inform patients that they should store tobramycin ampules in a refrigerator. However, when they don't have a refrigerator available (e.g., transporting the medication), they may store the foil pouches (opened or unopened) at room temperature (up to 77°F) for up to 28 days.
- Advise patients that tobramycin is light sensitive; unopened ampules should be returned to the foil pouch. Avoid exposing the ampules to intense light. Unrefrigerated tobramycin, which is normally colorless to pale yellow, may darken with age; however, the color change does not indicate any change in the quality of the product.
- Inform patients that they should not use tobramycin is it is cloudy, if there are particles in the solution, or if it has been stored at room temperature for more than 28 days. They should not use tobramycin beyond the expiration date stamped on the ampule.
Indications
Dosing (Adult)
Dosing (Pediatric)
Dosage Forms
Warnings
Adverse Reactions
Overdose
Drug Interactions
Special Populations
Breasfeeding
Chemical Structure
Mechanism of Action
Pharmacokinetics
Counseling Points
MESH Terms & Keywords
|
|---|
|





