Propranolol (Inderal LA): Drug Monograph
|
|---|
- Management of hypertension alone or in combination with other antihypertensive agents, particularly a thiazide diuretic. Not indicated in the management of hypertensive emergencies
- To decrease angina frequency and increase exercise tolerance in patients with angina pectoris due to coronary atherosclerosis
- For the prophylaxis of common migraine headache. Not indicated for the treatment of a migraine attack
- Improvement of NYHA functional class in symptomatic patients with hypertrophic subaortic stenosis
- General Dosing & Administration Notes:
-
Long acting capsules should not be considered a simple mg-for-mg substitute for conventional propranolol. Retitration may be necessary.
-
Hypertension:
-
80 mg by mouth once daily
-
May increase to 120 mg once daily or higher until adequate blood pressure control is achieved
-
Maintenance: 120-160 mg daily; 640 mg/day may be required
-
Angina pectoris:
-
80 mg by mouth once daily
-
Increase gradually at 3- to 7-day intervals until optimal response is obtained
-
Maintenance: 160 mg daily
-
Maximum: 320 mg daily
-
Reduce dose gradually over a period of a few weeks if therapy is to be discontinued
-
Migraine:
-
80 mg by mouth once daily
-
Usual: 160-240 mg once daily
-
Discontinue gradually if a satisfactory response is not obtained within 4-6 weeks after reaching maximal dose
-
Hypertrophic subaortic stenosis:
-
80-160 mg by mouth once daily
- Exacerbation of angina and, in some cases, myocardial infarction, following abrupt discontinuance of propranolol therapy.
- When discontinuance is planned, the dosage should be gradually reduced over at least a few weeks, and the patient should be cautioned against interruption or cessation of therapy without the physician's advice.
- Since coronary artery disease may be unrecognized, it may be prudent to follow the above advice in patients considered at risk of having occult atherosclerotic heart disease who are given propranolol for other indications.
- Cardiogenic shock
- Sinus bradycardia and greater than first-degree block
- Bronchial asthma
- Patients with known hypersensitivity to propranolol hydrochloride
- Hypersensitivity and skin reactions - including anaphylactic/anaphylactoid reactions, cutaneous reactions, including Stevens-Johnson syndrome, toxic epidermal necrolysis, exfoliative dermatitis, erythema multiforme, and urticaria are possible.
- Cardiac failure - in patients with a history of heart failure, continued use of beta-blockers can, in some cases, lead to cardiac failure
- Nonallergic bronchospasm (e.g., chronic bronchitis, emphysema) - In general, patients with bronchospastic lung disease should not receive beta-blockers. Administer with caution in this setting since it ay provoke a bronchial asthmatic attack.
- Major surgery - chronically administered beta-blocking therapy should not be routinely withdrawn prior to major surgery; however, the impaired ability of the heart to respond to reflex adrenergic stimuli may augment the risks of general anesthesia and surgical procedures.
- Diabetes and hypoglycemia - may prevent the appearance of certain premonitory signs and symptoms (pulse rate and pressure changes) of acute hypoglycemia, especially in labile insulin-dependent diabetics. Propranolol therapy, particularly when given to infants and children, diabetic or not, has been associated with hypoglycemia especially during fasting as in preparation for surgery. Hypoglycemia has been reported in patients taking propranolol after prolonged physical exertion and in patients with renal insufficiency.
- Thyrotoxicosis - may mask certain clinical signs of hyperthyroidism. Abrupt withdrawal of treatment may be followed by an exacerbation of symptoms of hyperthyroidism, including thyroid storm. Propranolol may change thyroid-function tests, increasing T4 and reverse T3, and decreasing T3.
- Wolff-Parkinson-White syndrome - patients with this syndrome and tachycardia may require treatment with a pacemaker.
- Mild impaired hepatic or renal function - use with caution
- Intraocular pressure - may can reduction in pressure. May interfere with the glaucoma screening test. Withdrawal may lead to a return of increased intraocular pressure
- History of severe anaphylactic reaction to a variety of allergens - patients may be more reactive to repeated challenge, accidental, diagnostic, or therapeutic. Patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction.
- Bradycardia
- Congestive heart failure
- Intensification of AV block
- Hypotension
- Paresthesia of hands
- Thrombocytopenic purpura
- Arterial insufficiency, usually of the Raynaud type
- Light-headedness
- Mental depression manifested by insomnia, lassitude, weakness, fatigue
- Catatonia
- Visual disturbances
- Hallucinations
- Vivid dreams
- An acute reversible syndrome characterized by disorientation for time and place, short-term memory loss, emotional lability, slightly clouded sensorium, and decreased performance on neuropsychometrics
- Nausea, vomiting
- Epigastric distress
- Abdominal cramping, diarrhea
- Constipation
- Mesenteric arterial thrombosis
- Ischemic colitis
- Hypersensitivity reactions, including anaphylactic/anaphylactoid reactions
- Pharyngitis and agranulocytosis
- Erythematous rash
- Fever combined with aching and sore throat
- Laryngospasm
- Respiratory distress
- Bronchospasm
- Agranulocytosis, nonthrombocytopenic purpura
- Thrombocytopenic purpura
- Systemic lupus erythematosus
- Stevens-Johnson syndrome
- Toxic epidermal necrolysis
- Dry eyes
- Exfoliative dermatitis
- Erythema multiforme
- Urticaria
- Alopecia
- SLE-like reactions
- Psoriassiform rashes
- Male impotence
- Peyronie's disease
- Not significantly dialyzable
- Evacuate gastric contents, taking care to prevent pulmonary aspiration
- Use supportive therapy for hypotension and bradycardia - may use glucagon (50-150 mcg/kg, intravenously followed by continuous drip of 1-5 mg/hour for positive chronotropic effect
- Isoproterenol, dopamine or phosphodiesterase inhibitors may also be useful
- No epinephrine
- Atropine or isoproterenol for bradycardia
- Drugs that affect CYP2D6, 1A2, or 2C19 metabolic pathways - may lead to clinically relevant drug interactions and changes on its efficacy and/or toxicity
- Alcohol - may increase plasma levels of propranolol
- Antiarrhythmics:
- Propafenone has negative inotropic and beta-blocking properties that can be additive to those of propranolol
- Quinidine - increases the concentration of propranolol and produces greater degrees of clinical beta-blockade and may cause postural hypotension
- Amiodarone - negative chronotropic properties may be additive to those seen with β-blockers
- Lidocaine - clearance is reduced. Possible lidocaine toxicity
- Drugs that slow A-V nodal conduction (e.g., lidocaine and calcium channel blockers) - use caution when administering
- Digitalis Glycosides - concomitant use can increase the risk of bradycardia
- Calcium channel blockers - may depress myocardial contractility or atrioventricular conduction
- Verapamil and beta-blockers - significant bradycardia, heart failure, and cardiovascular collapse possible
- Diltiazem - in patients with cardiac disease, possible bradycardia, hypotension, high degree heart block, and heart failure.
- ACE inhibitors - can cause hypotension, particularly in the setting of acute myocardial infarction.
- Clonidine - administer cautiously
- Prazosin - prolongation of first dose hypotension may be possible
- Terazosin or doxazosin - postural hypotension possible
- Reserpine - closely observe patients for excessive reduction of resting sympathetic nervous activity, which may result in hypotension, marked bradycardia, vertigo, syncopal attacks, or orthostatic hypotension
- Inotropic agents - patients on long-term therapy with propranolol may experience uncontrolled hypertension if administered epinephrine as a consequence of unopposed alpha-receptor stimulation. Epinephrine is not indicated in the treatment of propranolol overdose.
- Isoproterenol and dobutamine - propranolol's effects can be reversed by administration of such agents. Propranolol may reduce sensitivity to dobutamine stress echocardiography in patients undergoing evaluation for myocardial ischemia.
- NSAIDs - may blunt the antihypertensive effect of propranolol
- Indomethacin - may reduce the efficacy of propranolol in reducing blood pressure and heart rate
- MAO inhibitors or tricyclic antidepressants - hypotensive effects may be exacerbated
- Methoxyflurane and trichloroethylene - may depress myocardial contractility
- Warfarin - increases the concentration of warfarin. Prothrombin time should be monitored.
- Haloperidol - hypotension and cardiac arrest possible
- Pregnancy: Pregnancy Category C
- Labor and Delivery: None
- Nursing Mothers: Propranolol is excreted in human milk. Caution should be exercised when administered to a nursing woman.
- Renal Impairment: None
- Hepatic Impairment: None
- Pediatric Patients: Safety and effectiveness have not been established. Bronchospasm and congestive heart failure have been reported with propranolol therapy in pediatric patients.
- Geriatric Patients: In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of the decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
- Propranolol is excreted in human milk. Caution should be exercised when administered to a nursing woman.
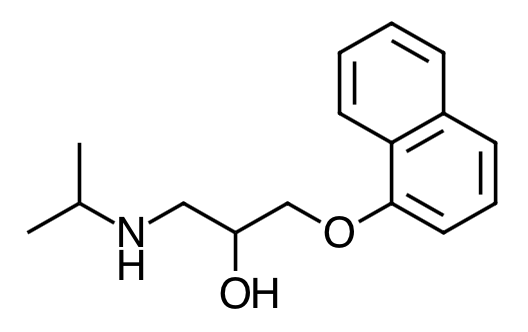
- Scientific Name: 2-Propanol, 1-[(1-methylethyl)amino]-3-(1-naphthalenyloxy)-, hydrochloride,(±)-
- Empirical Formula: C16H21NO2.HCl
- Molecular Weight: 295.80
-
Pharmacology Info:
-
Propranolol is a nonselective, beta-adrenergic receptor-blocking agent possessing no other autonomic nervous system activity. It specifically competes with beta-adrenergic receptor-stimulating agents for available receptor sites. When access to beta-receptor sites is blocked by propranolol, the chronotropic, inotropic, and vasodilator responses to beta-adrenergic stimulation are decreased proportionately. At dosages greater than required for beta blockade, propranolol also exerts a quinidine-like or anesthetic-like membrane action, which affects the cardiac action potential. The significance of the membrane action in the treatment of arrhythmias is uncertain.
-
Propranolol LA should not be considered a simple mg-for-mg substitute for conventional propranolol and the blood levels achieved do not match (are lower than) those of two to four times daily dosing with the same dose. When changing to long acting from conventional propranolol, a possible need for retitration upwards should be considered, especially to maintain effectiveness at the end of the dosing interval. In most clinical settings, however, such as hypertension or angina where there is little correlation between plasma levels and clinical effect, long acting has been therapeutically equivalent to the same mg dose of conventional propranolol as assessed by 24-hour effects on blood pressure and on 24-hour exercise responses of heart rate, systolic pressure, and rate pressure product.
- The mechanism of the antihypertensive effect of propranolol has not been established. Among the factors that may be involved in contributing to the antihypertensive action include: (1) decreased cardiac output, (2) inhibition of renin release by the kidneys, and (3) diminution of tonic sympathetic nerve outflow from vasomotor centers in the brain. Although total peripheral resistance may increase initially, it readjusts to or below the pretreatment level with chronic use of propranolol. Effects of propranolol on plasma volume appear to be minor and somewhat variable.
- In angina pectoris, propranolol generally reduces the oxygen requirement of the heart at any given level of effort by blocking the catecholamine-induced increases in the heart rate, systolic blood pressure, and the velocity and extent of myocardial contraction. Propranolol may increase oxygen requirements by increasing left ventricular fiber length, and diastolic pressure, and systolic ejection period. The net physiologic effect of beta-adrenergic blockade is usually advantageous and is manifested during exercise by delayed onset of pain and increased work capacity.
- Propranolol exerts its antiarrhythmic effects in concentrations associated with beta-adrenergic blockade, and this appears to be its principal antiarrhythmic mechanism of action. In dosages greater than required for beta blockade, propranolol also exerts a quinidine-like or anesthetic-like membrane action, which affects the cardiac action potential. The significance of the membrane action in the treatment of arrhythmias is uncertain.
- The mechanism of the anti-migraine effect of propranolol has not been established. Beta-adrenergic receptors have been demonstrated in the pail vessels of the brain.
- In a retrospective, uncontrolled study, 107 patients with diastolic blood pressure 110 to 150 mmHg received propranolol 120 mg tid for at least 6 months, in addition to diuretics and potassium, but with no other hypertensive agent. Propranolol contributed to control of diastolic blood pressure, but the magnitude of the effect of propranolol on blood pressure cannot be ascertained.
- Four double-blind, randomized, crossover studies were conducted in a total of 74 patients with mild or moderately severe hypertension treated with long acting 160 mg once daily or propranolol 160 mg given either once daily or in two 80 mg doses. Three of these studies were conducted over a 4-week treatment period. One study was assessed after a 24-hour period. The long acting propranolol was as effective as propranolol in controlling hypertension (pulse rate, systolic and diastolic blood pressure) in each of these trials.
- Angina Pectoris: In a double-blind, placebo-controlled study of 32 patients of both sexes, aged 32 to 69 years, with stable angina, propranolol 100 mg tid was administered for 4 weeks and shown to be more effective than placebo in reducing the rate of angina episodes and in prolonging total exercise time.
- Twelve male patients with moderately severe angina pectoris were studied in a double-blind, crossover study. Patients were randomized to either long acting 160 mg daily or conventional propranolol 40 mg four times a day for 2 weeks. Nitroglycerine tablets were allowed during the study. Blood pressure, heart rate and ECGs were recorded during serial exercise treadmill testing. The long acting was as effective as conventional propranolol for exercise heart rate, systolic and diastolic blood pressure, duration of angina pain and St-segment depression before or after exercise, exercise duration, angina attack rate and nitroglycerine consumption.
- In another double-blind, randomized, crossover trial, the effectiveness of propranolol long acting 160 mg daily and conventional propranolol 40 mg four times a day were evaluated in 13 patients with angina. ECGs were recorded while patients exercised until angina developed. The long acting was as effective as conventional propranolol for amount of exercise performed, ST-segment depression, number of angina attacks, amount of nitroglycerine consumed, systolic and diastolic blood pressures and heart rate at rest and after exercise.
- Migraine: In a 34-week, placebo-controlled, 4-period, dose-finding crossover study with a double-blind randomized treatment sequence, 62 patients with migraine received propranolol 2o to 89 mg 3 or 4 times daily. The headache unit index, a composite of the number of days with headache and the associated severity of the headache, was significantly reduced for patients receiving propranolol as compared to those on placebo.
- Hypertrophic Subaortic Stenosis: In an uncontrolled series of 13 patients with New York Heart Association (NYHA) class 2 or 3 symptoms and hypertrophic subaortic stenosis diagnosed at cardiac catheterization, oral propranolol 40-80 mg tid was administered and patients were followed for up to 17 months. Propranolol was associated with improved NYHA class for most patients.
- Absorption: Propranolol is highly lipophilic and almost completely absorbed after oral administration. However, it undergoes high first pass metabolism by the liver and on average, only about 25% of propranolol reaches the systemic circulation. The long acting capsules release propranolol HCL at a controlled and predictable rate. Peak blood levels following dosing occur at about 6 hours. The effect of food on bioavailability has not been investigated.
- Distribution: Approximately 90% of circulating propranolol is bound to plasma proteins (albumin and alpha-12 acid glycoprotein). The binding is enantiomer-selective. The S(-) enantiomer is preferentially bound to alpha-1 glycoprotein and the R(+) enantiomer preferentially bound to albumin. The volume of distribution of propranolol is approximately 4 liters/kg. Propranolol crosses the blood-brain barrier and the placenta, and is distributed into breast milk.
- Metabolism: Propranolol is extensively metabolized with most metabolites appearing in the urine. Propranolol is metabolized through three primary routes: aromatic hydroxylation (mainly 4-hydroxylation), N-dealkylation followed by further side-chain oxidation, and direct glucuronidation. It has been estimated that the percentage contributions of these routes to total metabolism are 42%, 41%, and 17%, respectively, but with considerable variability between individuals. The four major metabolites are propranolol glucuronide, naphthyloxylactic acid, and glucuronic acid, and sulfate conjugates of 4-hydroxy propranolol.
- In-vitro studies have indicated that the aromatic hydroxylation of propranolol is catalyzed mainly by polymorphic CYP2D6. Side-chain oxidation is mediated mainly by CYP1A2 and to some extent by CYP2D6. 4-hydroxy propranolol is a weak inhibitor of CYP2D6.
- Propranolol is also a substrate of CYP2C19 and a substrate for the intestinal efflux transporter, p-glycoprotein (p-gp). Studies suggest however that p-gp is not dose-limiting for intestinal absorption of propranolol in the usual therapeutic dose range.
- In healthy subjects, no difference was observed between CYP2D6 extensive metabolizers (EMs) and poor metabolizers (PMs) with respect to oral clearance or elimination half-life. Partial clearance of 4-hydroxy propranolol was significantly higher and naphthyloxylactic acid was significantly lower in Ems than PMs.
- When measured at steady state over a 24-hour period the areas under the propranolol plasma concentration-time curve (AUCs) for the long acting capsules are approximately 60% to 65% of the AUCs for a comparable divided daily dose of conventional tablets. The lower AUCs for the long acting capsules are due to greater hepatic metabolism of propranolol, resulting from the slower rate of absorption of propranolol. Over a twenty-four (24) hour period, blood levels are fairly constant for about twelve (12) hours, then decline exponentially. The apparent plasma half-life is about 10 hours.
- Elimination: T1/2 = 10 hours
- Special Populations
- Geriatric: The pharmacokinetics of the long acting capsule have not been investigated in patients over 65 years of age. In a study of 12 elderly (62-79 years old) and 12 young (25-33 years old) healthy subjects, the clearance of S-enantiomer of propranolol was decreased in the elderly. Additionally, the half-life of both the R- and S-propranolol were prolonged in the elderly compared with the young (11 hours vs. 5 hours). Clearance of propranolol is reduced with aging due to decline in oxidation capacity (ring oxidation ad side chain oxidation). Conjugation capacity remains unchanged. In a study of 32 patients age 30 to 84 years given a single 20 mg dose of propranolol an inverse correlation was found between age and the partial metabolic clearances to 4-hydroxypropranolol (40HP ring oxidation) and to naphthoxylactic acid (NLA-side chain oxidation). No correlation was found between age and the partial metabolic clearance to propranolol glucuronide (PPLG conjugation).
- Gender: In a study of 9 healthy women and 12 healthy men, neither the administration of testosterone nor the regular course of the menstrual cycle affected the plasma binding of the propranolol enantiomers. In contrast, there was a significant, although non-enantioselective diminution of the binding of propranolol after treatment with ethinyl estradiol. These findings are inconsistent with another study, in which administration of testosterone cypionate confirmed the stimulatory role of this hormone on propranolol metabolism and concluded that the clearance of propranolol in men is dependent on circulating concentrations of testosterone. In women, none of the metabolic clearances for propranolol showed any significant association with either estradiol or testosterone.
- Race: A study conducted in 12 Caucasian and 13 African-American male subjects taking propranolol, showed that at stead state, the clearance of R(+) and S(-) propranolol were about 76% and 53% higher in African-Americans than in Caucasians, respectively.
- Chinese subjects had a greater proportion (18% to 45% higher) of unbound propranolol in plasma compared to Caucasians, which was associated with a lower plasma concentration of alpha 1-acid glycoprotein.
- Renal Insufficiency: The pharmacokinetics of long acting propranolol have not been investigated in patients with renal insufficiency.In a study conducted in 5 patients with chronic renal failure, 6 patients on regular dialysis, and 5 healthy subjects, who received a single oral dose of 40 mg of propranolol, the peak plasma concentrations (Cmax) of propranolol in the chronic renal failure group were 2 to 3-fold higher (161±41 ng/mL) than those observed in the dialysis patients (47±9 ng/mL) and in the healthy subjects (26±1 ng/mL). Propranolol plasma clearance was also reduced in the patients with chronic renal failure. Studies have reported a delayed absorption rate and a reduced half-life of propranolol in patients with renal failure of varying severity. Despite this shorter plasma half-life, propranolol peak plasma levels were 3-4 times higher and total plasma levels of metabolites were up to 3 times higher in these patients than in subjects with normal renal function. Chronic renal failure has been associated with a decrease in drug metabolism via down regulation of hepatic cytochrome P450 activity resulting in a lower "first-pass" clearance. Propranolol is not significantly dialyzable.
- Hepatic Insufficiency: The pharmacokinetics of long acting propranolol have not been investigated in patients with hepatic insufficiency. Propranolol is extensively metabolized by the liver. In a study conducted in 6 patients with cirrhosis and 7 healthy subjects receiving 160 mg of a long acting preparation of propranolol once a day for 7 days, the steady-state propranolol concentration in patients with cirrhosis was increased 2.5-fold in comparison to controls. In the patients with cirrhosis, the half-life obtained after a single intravenous dose of 10 mg propranolol increased to 7.2 hours compared to 2.9 hours in control.
- Drug Interactions: All drug interaction studies were conducted with propranolol. There are no data on drug interactions with the long acting capsules.
- Interactions with substrates, inhibitors or inducers of Cytochrome P-450 enzymes: Because propranolol's metabolism involves multiple pathways in the Cytochrome P-450 system (CYP2D6), 1A2, 2C19), co-administration with drugs that are metabolized by, or affect the activity (induction or inhibition) of one or more of these pathways may lead to clinically relevant drug interactions.
- Substrates or inhibitors of CYP2D6: Blood levels and/or toxicity of propranolol may be increased by co-administration with substrates or inhibitors of CYP2D6, such as amiodarone, cimetidine, delavudin, fluoxetine, paroxetine, quinidine, and ritonavir. No interactions were observed with either ranitidine or lansoprazole.
- Substrates or inhibitors of CYP1A2: Blood levels and/or toxicity of propranolol may be increased by co-administration with substrates or inhibitors of CYP1A2, such as imipramine, cimetidine, ciprofloxacin, fluvoxamine, isoniazid, ritonavir, theophylline, zileuton, zolmitriptan, and rizatriptan.
- Substrates or inhibitors of CYP2C19: Blood levels and/or toxicity of propranolol may be increased by co-administration with substrates or inhibitors of CYP2C19, such as fluconazole, cimetidine, fluoxetine, fluvoxamine, tenioposide, and tolbutamide. No interaction was observed with omeprazole.
- Inducers of hepatic drug metabolism: Blood levels of propranolol may be decreased by co-administration with inducers such as rifampin, ethanol, phenytoin, and phenobarbital. Cigarette smoking also induces hepatic metabolism and has been shown to increase up to 77% the clearance of propranolol, resulting in decreased plasma concentrations.
- Cardiovascular Drugs
- Antiarrhythmics: The AUC of propafenone is increased by more than 200% by co-administration of propranolol. The metabolism of propranolol is reduced by co-administration of quinidine, leading to a two to three fold increased blood concentration and greater degrees of clinical beta-blockade.
- The metabolism of lidocaine is inhibited by co-administration of propranolol, resulting in a 25% increase in lidocaine concentrations.
- Calcium Channel Blockers: The mean Cmax and AUC of propranolol are increased respectively, by 50% and 30% by co-administration of nisoldipine and by 80% and 47% by co-administration of nicardipine. The mean Cmax and AUC of nifedipine are increased by 64% and 79%, respectively, by co-administration of propranolol. Propranolol does not affect the pharmacokinetics of verapamil and norverapamil. Verapamil does not affect the pharmacokinetics of propranolol.
- Non-Cardiovascular Drugs
- Migraine Drugs: Administration of zolmitriptan or rizatriptan with propranolol resulted in increased concentrations of zolmitriptan (AUC increased by 56% and Cmax by 37%) or rizatriptan (the AUC and Cmax were increased by 67% and 75%, respectively).
- Theophylline: Co-administration of theophylline with propranolol decreased theophylline oral clearance by 30% to 52%.
- Benzodiazepines: Propranolol can inhibit the metabolism of diazepam, resulting in increased concentrations of diazepam and its metabolites. Diazepam does not alter the pharmacokinetics of propranolol. The pharmacokinetics of oxazepam, triazolam, lorazepam, and alprazolam are not affected by co-administration of propranolol.
- Neuroleptic Drugs: Co-administration of long-acting propranolol at doses greater than or equal to 160 mg/day resulted in increased thioridazine plasma concentrations ranging from 55% to 369% and increased thioridazine metabolite (mesoridazine) concentrations ranging from 33% to 209%. Co-administration of chlorpromazine with propranolol resulted in a 70% increase in propranolol plasma level.
- Anti-Ulcer Drugs: Co-administration of propranolol with cimetidine, a non-specific CYP450 inhibitor, increased propranolol AUC and Cmax by 46% and 35%, respectively. Co-administration with aluminum hydroxide gel (1200 mg) may result in a decrease in propranolol concentrations. Co-administration of metoclopramide with the long0acting propranolol did not have a significant effect on propranolol's pharmacokinetics.
- Lipid Lowering Drugs: Co-administration of cholestyramine or colestipol with propranolol resulted I up to 50% decrease in propranolol concentrations. Co-administration of propranolol with lovastatin or pravastatin, decreased 18% to 23%, the AUC of both, but did not alter their pharmacodynamics. Propranolol did not have an effect on the pharmacokinetics of fluvastatin.
- Warfarin: Concomitant administration of propranolol and warfarin has been shown to increase warfarin bioavailability and increase prothrombin time.
Indications
Dosing (Adult)
Black Box Warnings
Contraindications
Warnings
Adverse Reactions
Overdose
Drug Interactions
Special Populations
Breasfeeding
Chemical Structure
Mechanism of Action
Pharmacodynamics
Pharmacokinetics
MESH Terms & Keywords
|
|---|
|





