Pramipexole (Mirapex, Mirapex ER): Drug Monograph
|
|---|
- Treatment of Parkinson's disease (Immediate and extended release)
- Treatment of moderate to severe primary restless legs syndrome (Immediate release)
- General Dosing & Administration Notes:
- Take with or without food
- Extended release: Swallow whole; do not chew, crush, or divide tablet
- If a significant interruption in therapy has occurred, retitration may be warranted
- Parkinson's disease, immediate release (IR) tablets:
- 0.125 mg by mouth three times a day
-
May increase gradually not more frequently than every 5-7 days (e.g., Week 2: 0.25 mg three times a day; Week 3: 0.5 mg three times a day; Week 4: 0.75 mg three times a day; Week 5: 1 mg three times a day; Week 6: 1.25 mg three times a day; Week 7: 1.5 mg three times a day)
-
Maintenance: 0.5-1.5 mg three times a day in equally divided doses with or without levodopa
-
Parkinson's disease, extended release (ER) tablets:
-
0.375 mg once daily
-
Based on efficacy and tolerability, may increase gradually but not more frequently than every 5-7 days, 1st to 0.75 mg/day then by 0.75 mg increments
-
Maximum: 4.5 mg/day
- Conversions:
-
Switching from immediate release tablets to extended release: May switch overnight at the same daily dose; monitor to determine if dosage adjustment is necessary
-
Restless legs syndrome, primary, moderate to severe:
-
0.125 mg once daily to be administered 2 - 3 hours before bedtime
-
May increase dose every 4-7 days up to 0.5 mg/day, if needed
-
Titration Step 1: 0.125 mg once daily
-
Titration Step 2: 0.25 mg once daily
-
Titration Step 3: 0.5 mg once daily
- Discontinuation, Parkinson's disease:
-
Taper off at a rate of 0.75 mg daily until daily dose has been reduced to 0.75 mg, then reduce dose by .375 mg/day
- Parkinson's disease:
-
Mild, CrCl >50 mL/min:
-
0.125 mg three times a day; maximum: 1.5 mg three times a day
-
Moderate, CrCl 30-50 mL/min (IR tablets):
-
0.125 mg twice daily; maximum: 0.75 mg three times a day
-
Moderate, CrCl 30-50 mL/min (ER tablets):
-
Administer every other day; carefully assess therapeutic response and tolerability before increasing to daily dosing after 1 week, and before any additional titration in 0.375 mg increments. Dose adjustment should occur no more frequently than at weekly intervals. Maximum: 2.25 mg/day
-
Severe, CrCl <30 mL/min (IR tablets):
-
0.125 mg once daily; maximum: 1.5 mg once daily
-
Severe, CrCl <30 mL/min/Hemodialysis (ER tablets)
-
Not recommended
- Restless legs syndrome:
-
Moderate and severe, CrCl 20-60 mL/min: Increase duration between titration steps to 14 days
- Tablets, immediate release: 0.125 mg, 0.25 mg, 0.5 mg, 0.75 mg, 1 mg, 1.5 mg
- Tablets, extended release: 0.375 mg, 0.75 ng, 1.5 mg, 2.25 mg, 3 mg. 3.75 mg, 4.5 m
- Falling asleep during activities of daily living - sudden onset of sleep may occur without warning; advise patients to report symptoms
- Symptomatic orthostatic hypotension - monitor during dose escalation
- Impulse control/compulsive behaviors/intense urges
- Hallucinations and psychotic-like behavior - risk increases with age
- Dyskinesia - may be caused or exacerbated by pramipexole
- Events reported with dopaminergic therapy which include hyperpyrexia and confusion, fibrotic complications, and melanoma
- Early PD without levodopa:
- Nausea
- Dizziness
- Somnolence
- Insomnia
- Constipation
- Asthenia
- Hallucinations
- Advanced PD with levodopa:
- Postural (orthostatic) hypotension
- Dyskinesia
- Extrapyramidal syndrome
- Insomnia
- Dizziness
- Hallucinations
- Accidental injury
- Dream abnormalities
- Confusion
- Constipation
- Asthenia
- Somnolence
- Dystonia
- Gait abnormality
- Hypertonia
- Dry mouth
- Amnesia
- Urinary frequency
- RLS:
- Nausea
- Somnolence
- Fatigue
- Headache
- There is no clinical experience with significant overdosage.
- Pulse rate may increase.
- If signs of CNS stimulation are present, a phenothiazine or other butyrophenone neuroleptic agent may be indicated.
- Management may require general supportive measures along with gastric lavage, IV fluids, and electrocardiogram monitoring.
- Dopamine antagonists - neuroleptics (phenothiazines, butyrophenones, thioxanthenes) or metoclopramide, may diminish the effectiveness of pramipexole
- Pregnancy: Pregnancy Category C
- Labor and Delivery: None
- Nursing Mothers: Treatment will result in an inhibition of prolactin secretion in humans and rats. It is not known whether this drug is excreted in human milk. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
- Renal Impairment: Clearance is extremely low in dialysis patients. Caution should be exercised when administering to patients with renal disease. Dosage adjustment necessary.
- Hepatic Impairment: None
- Pediatric Patients: Safety and effectiveness has not been established.
- Geriatric Patients: Total oral clearance of pramipexole is approximately 30% lower in subjects older than 65 years compared with younger subjects, which results in an increase in elimination half-life from approximately 8.5 hours to 12 hours. No apparent differences in efficacy or safety between older and younger PD or RLS patients, except that the relative risk of hallucination association with the use of pramipexole is increased in the elderly.
- Treatment will result in an inhibition of prolactin secretion in humans and rats. It is not known whether this drug is excreted in human milk. A decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
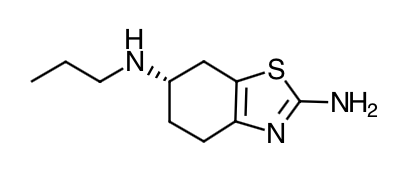
- Scientific Name: (S)-2-amino-4,5,6,7-tetrahydro-6-(propylamino)benzothiazole dihydrochloride monohydrate
- Empirical Formula: C10H17N3S•2HCl•H2O
- Molecular Weight: 302.26
- Pramipexole is a non-ergot dopamine agonist with high relative in vitro specificity and full intrinsic activity at the D2 subfamily of dopamine receptors, binding with higher affinity to D3 than to D2 or D4 receptor subtypes.
- Parkinson's disease: The precise mechanism of action of pramipexole as a treatment for Parkinson's disease is unknown, although it is believed to be related to its ability to stimulate dopamine receptors in the striatum. This conclusion is supported by electrophysiologic studies in animals that have demonstrated that pramipexole influences striatal neuronal firing rates via activation of dopamine receptors in the striatum and the substantia nigra, the site of neurons that send projections to the striatum. The relevance of D3 receptor binding in Parkinson's disease is unknown.
- Restless Legs Syndrome (RLS): The precise mechanism of action of pramipexole as a treatment for RLS is unknown. Although the pathophysiology of RLS is largely unknown, neuropharmacological evidence suggests primary dopaminergic system involvement. Positron Emission Tomographic (PET) studies suggest that a mild striatal presynaptic dopaminergic dysfunction may be involved in the pathogenesis of RLS.
- The effect of pramipexole on the QT interval of the ECG was investigated in a clinical study in 60 healthy male and female volunteers. All subjects initiated treatment with 0.375 mg extended release pramipexole tablets administered once daily, and were up-titrated every 3 days to 2.25 mg and 4.5 mg daily, a faster rate of titration than recommended in the label. No dose- or exposure-related effect on mean QT intervals was observed; however the study did not have a valid assessment of assay sensitivity. The effect of pramipexole on QTc intervals at higher exposures achieved either due to drug interactions (e.g., with cimetidine), renal impairment, or at higher doses has not been systematically evaluated.
- Although mean values remained within normal reference ranges throughout the study, supine systolic blood pressure (SBP), diastolic blood pressure (DBP), and pulse rate for subjects treated with pramipexole generally increased during the rapid up-titration phase, by 10 mmHg, 7 mmHg, and 10 bpm higher than placebo, respectively. Higher SBP, DBP, and pulse rates compared to placebo were maintained until the pramipexole doses were tapered; values on the last day of tapering were generally similar to baseline values. Such effects have not been observed in clinical studies with Parkinson's disease patients, who were titrated according to labeled recommendations.
- Absorption: Pramipexole is rapidly absorbed, reaching peak concentrations in approximately 2 hours. The absolute bioavailability of pramipexole is greater than 90%, indicating that it is well absorbed and undergoes little presystemic metabolism. Food does not affect the extent of pramipexole absorption, although the time of maximum plasma concentration Tmax is increased by about 1 hour when the drug is taken with a meal. Increase in systemic exposure of pramipexole following oral administration of 0.375 mg to 4.5 mg of extended release tablets was dose-proportional and steady state of exposure is reached within 5 days of continuous dosing. Relative bioavailability of extended release tablets compared with immediate-release tablets was approximately 100%. In a repeat-dose study in healthy, normal volunteers, extended release tablets 4.5 mg administered once daily was bioequivalent with regard to Cmax and AUC over 24 hours to immediate-release pramipexole tablets 1.5 mg administered three times daily. The average time-to-peak concentration for extended release tablets is 6 hours. Administration of extended release tablets with food (i.e., high-fat meal) did not affect AUC but increased Cmax by approximately 20% and delayed Tmax by approximately 2 hours compared with dosing under fasted conditions; these differences are not considered to be clinically relevant.
- Distribution: Pramipexole is extensively distributed, having a volume of distribution of about 500 L (coefficient of variation [CV]=20%). It is about 15% bound to plasma proteins. Pramipexole distributes into red blood cells as indicated by an erythrocyte-to-plasma ratio of approximately 2.
- Metabolism: Pramipexole is metabolized only to a negligible extent (<10%). No specific active metabolite has been identified in human plasma or urine.
- Elimination: Urinary excretion is the major route of pramipexole elimination, with 90% of a pramipexole dose recovered in urine, almost all as unchanged drug. The renal clearance of pramipexole is approximately 400 mL/min (CV=25%), approximately three times higher than the glomerular filtration rate. Thus, pramipexole is secreted by the renal tubules, probably by the organic cation transport system.
- Specific Populations: Because therapy with pramipexole is initiated at a low dose and gradually titrated upward according to clinical tolerability to obtain the optimum therapeutic effect, adjustment of the initial dose based on gender, weight, race, or age is not necessary. However, renal insufficiency, which can cause a large decrease in the ability to eliminate pramipexole, may necessitate dosage adjustment.
- Gender: Pramipexole clearance is about 30% lower in women than in men, but this difference can be accounted for by differences in body weight. There is no difference in half-life between males and females.
- Age: Pramipexole clearance decreases with age as the half-life and clearance are about 40% longer and 30% lower, respectively, in elderly (aged 65 years or older) compared with young healthy volunteers (aged less than 40 years). This difference is most likely due to the reduction in renal function with age, since pramipexole clearance is correlated with renal function, as measured by creatinine clearance.
- Race: No racial differences in metabolism and elimination have been identified.
- Parkinson's Disease Patients: A cross-study comparison of data suggests that the clearance of pramipexole may be reduced by about 30% in Parkinson's disease patients compared with healthy elderly volunteers. The reason for this difference appears to be reduced renal function in Parkinson's disease patients, which may be related to their poorer general health. The pharmacokinetics of pramipexole were comparable between early and advanced Parkinson's disease patients.
- Restless Legs Syndrome Patients: A cross-study comparison of data suggests that the pharmacokinetic profile of pramipexole administered once daily in RLS patients is similar to the pharmacokinetic profile of pramipexole in healthy volunteers.
- Hepatic Impairment: The influence of hepatic insufficiency on pramipexole pharmacokinetics has not been evaluated. Because approximately 90% of the recovered dose is excreted in the urine as unchanged drug, hepatic impairment would not be expected to have a significant effect on pramipexole elimination.
- Renal Impairment: Clearance of pramipexole was about 75% lower in patients with severe renal impairment (creatinine clearance approximately 20 mL/min) and about 60% lower in patients with moderate impairment (creatinine clearance approximately 40 mL/min) compared with healthy volunteers. In patients with varying degrees of renal impairment, pramipexole clearance correlates well with creatinine clearance. Therefore, creatinine clearance can be used as a predictor of the extent of decrease in pramipexole clearance.
- Drug Interactions:
- Carbidopa/levodopa: Carbidopa/levodopa did not influence the pharmacokinetics of pramipexole in healthy volunteers (N=10). Pramipexole did not alter the extent of absorption (AUC) or the elimination of carbidopa/levodopa, although it caused an increase in levodopa Cmax by about 40% and a decrease in Tmax from 2.5 to 0.5 hours.
- Selegiline: In healthy volunteers (N=11), selegiline did not influence the pharmacokinetics of pramipexole.
- Amantadine: Population pharmacokinetic analyses suggest that amantadine may slightly decrease the oral clearance of pramipexole.
- Cimetidine: Cimetidine, a known inhibitor of renal tubular secretion of organic bases via the cationic transport system, caused a 50% increase in pramipexole AUC and a 40% increase in half-life (N=12).
- Probenecid: Probenecid, a known inhibitor of renal tubular secretion of organic acids via the anionic transporter, did not noticeably influence pramipexole pharmacokinetics (N=12).
- Other drugs eliminated via renal secretion: Population pharmacokinetic analysis suggests that coadministration of drugs that are secreted by the cationic transport system (e.g., cimetidine, ranitidine, diltiazem, triamterene, verapamil, quinidine, and quinine) decreases the oral clearance of pramipexole by about 20%, while those secreted by the anionic transport system (e.g., cephalosporins, penicillins, indomethacin, hydrochlorothiazide, and chlorpropamide) are likely to have little effect on the oral clearance of pramipexole. Other known organic cation transport substrates and/or inhibitors (e.g., cisplatin and procainamide) may also decrease the clearance of pramipexole.
- CYP interactions: Inhibitors of cytochrome P450 enzymes would not be expected to affect pramipexole elimination because pramipexole is not appreciably metabolized by these enzymes in vivo or in vitro. Pramipexole does not inhibit CYP enzymes CYP1A2, CYP2C9, CYP2C19, CYP2E1, and CYP3A4. Inhibition of CYP2D6 was observed with an apparent Ki of 30 μM, indicating that pramipexole will not inhibit CYP enzymes at plasma concentrations observed following the clinical dose of 4.5 mg/day (1.5 mg TID).
- Instruct patients to take pramipexole only as prescribed. If a dose is missed, advise patients not to double their net dose. If a dose is missed or the extended release tablets, the tablet should be taken as soon as possible, but no later than 12 hours after the regularly schedule time. After 12 hours, the missed dose should be skipped and the next dose should be taken on the following day at the regularly scheduled time. Tablets may be taken with or without food. If patients develop nausea, advise that taking tablets with food may reduce the occurrence of nausea. Ensure that patients do not take both extended-release along with regular tablets of pramipexole.
- Alert patients to the potential sedating effects associated with treatment, including somnolence and the possibility of falling asleep while engaged in activities of daily living. Patients should neither drive a car nor engage in other potentially dangerous activities until they have gained sufficient experience with the tablets to gauge whether or not they affect their mental and/or motor performance adversely. Advise caution when patients are taking other sedating medications or alcohol in combination with pramipexole and when taking concomitant medications that increase plasma levels of pramipexole (e.g., cimetidine).
- Alert patients and their caregivers to the possibility that they nay experience intense urges to spend money uncontrollably, intense urges to gamble, increased sexual urges, binge eating and/or other intense urges and the inability to control these urges while on treatment.
- Inform patients that hallucinations and other psychotic-like behavior can occur and that the elderly are at a higher risk than younger patients with Parkinson's disease.
- Advise patients that they may develop postural (orthostatic) hypotension, with or without symptoms such as dizziness, nausea, fainting or blackouts, and sometimes, sweating. Hypotension may occur more frequently during initial therapy. Caution patients against rising rapidly after sitting or lying down, especially if they have been doing so for prolonged periods and especially at the initiation of treatment.
- Advise women to notify their physicians if they become pregnant or intend to become pregnant during therapy.
- Advise women to notify their physicians if they intend to breastfeed or are breastfeeding an infant.
Indications
Dosing (Adult)
Renal Dosing
Dosage Forms
Warnings
Adverse Reactions
Overdose
Drug Interactions
Special Populations
Breasfeeding
Chemical Structure
Mechanism of Action
Pharmacodynamics
Pharmacokinetics
Counseling Points
MESH Terms & Keywords
|
|---|
|





