Gabapentin (Neurontin): Drug Monograph
|
|---|
- Post-herpetic neuralgia in adults
- Adjunctive therapy in the treatment of partial seizures, with and without secondary generalization, in adults and pediatric patients ≥3 years of age with epilepsy.
- Neuropathic pain (including Diabetic neuropathy):
- Not FDA approved indication
- 300 mg daily, but can be titrated up to 3600 mg/day in three divided doses
- Partial seizures:
- 300 mg three times a day
- Maintenance: 300-600 mg three times a day
- Doses up to 2400 mg/day (long-term) and 3600 mg/day (short-term) have been well tolerated. Administer in divided doses of three times a day using 300 mg or 400 mg capsules, or 600 mg or 800 mg tablets
- Dosing intervals generally should not exceed 12 hours
- Post-herpetic neuralgia:
- Single 300 mg dose on Day 1, 300 mg twice a day on Day 2, and 300 mg three times per day on Day 3
- May subsequently increase as needed up to 1800 mg/day (600 mg three times daily)
- Epileptic pediatric
patients age 3-12 years:
- Range from 10-15 mg/kg/day in 3 divided doses.
- Partial seizures:
- 3-11 years:
- 10-15 mg/kg/day in 3 divided doses.
- Increase to recommended maintenance dose over a period of approximately 3 days.
- 3-4 years:
- Maintenance: 40 mg/kg/day in 3 divided doses
- 5-11 years:
- Maintenance: 25-35 mg/kg/day in 3 divided doses
- Doses up to 50 mg/kg/day have been well tolerated
- Dosing intervals should not exceed 12 hours
- ≥12 years:
- 300 mg three times a day.
- Maintenance: 300-600 mg three times a day.
- Doses up to 2400 mg/day (long-term) and 3600 mg/day (short-term) have been well tolerated.
- Administer three times a day using 300 mg or 400 mg capsules, or 600 mg or 800 mg tablets.
- Dosing intervals should not exceed 12 hours.
- CrCl≥60 mL/min:
- 900-3600 mg/day in 3 divided doses
- CrCl>30-59 mL/min:
- 400-1400 mg/day in 2 divided doses
- CrCl>15-29 mL/min:
- 200-700 mg single daily dose
- CrCl 15 mL/min:
- 100-300 mg single daily dos
- CrCl <15 mL/min:
- Reduce daily dose in proportion to CrCl
- Hemodialysis:
- Dose adjustment is necessary
- Capsules: 100 mg, 300 mg, 400 mg
- Tablets: 300 mg, 600 mg, 800 mg
- Oral solution: 250 mg/5 mL (5 ml, 6 mL, 470 mL, 473 mL)
- Suicidal behavior and ideation
- Neuropsychiatric adverse events in pediatric patients 3-12
years of age who have epilepsy. May
include emotional lability (primarily behavioral problems); hostility,
including aggressive behaviors; thought disorder, including concentration
problems and change in school performance; and hyperkinesia, primarily
restlessness and hyperactivity.
- Withdrawal precipitated seizure, status epilepticus
- Tumorigenic Potential
- Sudden and unexplained death in patients with epilepsy
- Drug reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan hypersensitivity
- Double vision, slurred speech, drowsiness, lethargy, and diarrhea may occur.
- Removed by hemodialysis.
- Pregnancy: Pregnancy Category C
- Labor and Delivery: None
- Nursing Mothers: Gabapentin is secreted into human milk following oral administration. Effect on nursing infant is unknown.
- Renal Impairment: Dosage adjustment necessary.
- Hepatic Impairment: None
- Pediatric Patients: Safety and effectiveness of gabapentin in the management of post-herpetic neuralgia in pediatric patients have not been established. Effectiveness ad adjunctive therapy in the treatment of partial seizures in pediatric patients below the age of 3 years has not been established.
- Geriatric Patients: Larger treatment effect in patients 75 years of age and older. Age-related decrease in renal function may be involved. Dose selection should be cautious and dose should be adjusted based on creatinine clearance values.
- Gabapentin is secreted into human milk following oral administration.
- Effect on nursing infant is unknown.
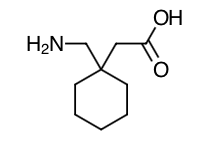
- Scientific Name: 1-(aminomethyl)cyclohexaneacetic acid
- Empirical Formula: C9H17NO2
- Molecular Weight: 171.24
- The mechanism by which gabapentin exerts its anticonvulsant action is unknown, but in animal test systems designed to detect anticonvulsant activity, gabapentin prevents seizures as do other marketed anticonvulsants.
- Gabapentin is structurally related to the neurotransmitter GABA (gamma-aminobutyric acid) but it does not modify GABA A or GABA B binding.
- It is also not metabolically converted into GABA or a GABA agonist and it is not an inhibitor of GABA uptake or degradation.
- Gabepentin was tested in radioligand binding assays at concentrations up to 100 mM and did not exhibit affinity for a number of other common receptor sites, including benzodiazepine, glutamate, N-methyl-D-aspartate (NMDA), quisqualate, kainate, strychnine-insensitive or strychnine-sensitive glycine, alpha 1, alpha 2, or beta adrenergic, adenosine A1 or A2, cholinergic muscarinic or nicotinic, dopamine D1 or D2, histamine H1, serotonin S1 or S2, opiate mu, delta or kappa, cannabinoid 1, voltage-sensitive calcium channel sites labeled with nitrendipine or diltiazem, or at voltage-sensitive sodium channel sites labeled with batrachotoxinin A 20-alpha-benzoate.
- Furthermore, gabapentin did not alter the cellular uptake of dopamine, noradrenaline, or serotonin.
- Absorption:
- Gabapentin bioavailability is not dose proportional; i.e., as dose is increased, bioavailability decreases.
- Bioavailability of gabapentin is approximately 60%, 47%, 34%, 33%, and 27% following 900, 1200, 2400, 3600, and 4800 mg/day given in 3 divided doses, respectively.
- Food has only a slight effect on the rate and extent of absorption of gabapentin (14% increase in AUC and Cmax).
- Distribution:
- The apparent volume of distribution is 58±6 L (mean ±SD).
- In patients with epilepsy, steady-state predose (Cmin) concentrations of gabapentin in cerebrospinal fluid were approximately 20% of the corresponding plasma concentrations.
- Protein Binding:
- Less than 3% of gabapentin circulates bound to plasma protein.
- Metabolism:
- Is not appreciably metabolized in humans.
- Elimination:
- Gabapentin is eliminated from the systemic circulation by renal excretion as unchanged drug.
- Gabapentin elimination half-life is 5 to 7 hours.
- In elderly patients, and in patients with impaired renal function, gabapentin plasma clearance is reduced.
- Gabapentin can be removed from plasma by hemodialysis. Dosage adjustment in patients with compromised renal function or undergoing hemodialysis is recommended.
- Special Populations
- Adult Patients With Renal Insufficiency: Subjects (N=60) with renal insufficiency (mean creatinine clearance ranging from 13-114 mL/min) were administered single 400 mg oral doses of gabapentin. The mean gabapentin half-life ranged from about 6.5 hours (patients with creatinine clearance >60 mL/min) to 52 hours (creatinine clearance <30 mL/min) and gabapentin renal clearance from about 90 mL/min (>60 mL/min group) to about 10 mL/min (<30 mL/min). Mean plasma clearance (CL/F) decreased from approximately 190 mL/min to 20 mL/min. Dosage adjustment in adult patients with compromised renal function is necessary. Pediatric patients with renal insufficiency have not been studied.
- Hemodialysis: In a study in anuric adult subjects (N=11), the apparent elimination half-life of gabapentin on nondialysis days was about 132 hours; during dialysis the apparent half-life of gabapentin was reduced to 3.8 hours. Hemodialysis thus has a significant effect on gabapentin elimination in anuric subjects. Dosage adjustment in patients undergoing hemodialysis is necessary.
- Hepatic Disease: Because gabapentin is not metabolized, no study was performed in patients with hepatic impairment.
- Age: The effect of age was studied in subjects 20-80 years of age. Apparent oral clearance (CL/F) of gabapentin decreased as age increased, from about 225 mL/min in those under 30 years of age to about 125 mL/min in those over 70 years of age. Renal clearance (CLr) and CLr adjusted for body surface area also declined with age; however, the decline in the renal clearance of gabapentin with age can largely be explained by the decline in renal function. Reduction of gabapentin dose may be required in patients who have age related compromised renal function.
- Pediatric: Gabapentin pharmacokinetics were determined in 48 pediatric subjects between the ages of 1 month and 12 years following a dose of approximately 10 mg/kg. Peak plasma concentrations were similar across the entire age group and occurred 2 to 3 hours post-dose. In general, pediatric subjects between 1 month and <5 years of age achieved approximately 30% lower exposure (AUC) than that observed in those 5 years of age and older. Accordingly, oral clearance normalized per body weight was higher in the younger children. Apparent oral clearance of gabapentin was directly proportional to creatinine clearance. Gabapentin elimination half-life averaged 4.7 hours and was similar across the age groups studied. A population pharmacokinetic analysis was performed in 253 pediatric subjects between 1 month and 13 years of age. Patients received 10 to 65 mg/kg/day given TID. Apparent oral clearance (CL/F) was directly proportional to creatinine clearance and this relationship was similar following a single dose and at steady state. Higher oral clearance values were observed in children <5 years of age compared to those observed in children 5 years of age and older, when normalized per body weight. The clearance was highly variable in infants <1 year of age. The normalized CL/F values observed in pediatric patients 5 years of age and older were consistent with values observed in adults after a single dose. The oral volume of distribution normalized per body weight was constant across the age range. These pharmacokinetic data indicate that the effective daily dose in pediatric patients with epilepsy ages 3 and 4 years should be 40 mg/kg/day to achieve average plasma concentrations similar to those achieved in patients 5 years of age and older receiving gabapentin at 30 mg/kg/day.
- Gender: Although no formal study has been conducted to compare the pharmacokinetics of gabapentin in men and women, it appears that the pharmacokinetic parameters for males and females are similar and there are no significant gender differences.
- Race: Pharmacokinetic differences due to race have not been studied. Because gabapentin is primarily renally excreted and there are no important racial differences in creatinine clearance, pharmacokinetic differences due to race are not expected.
- Instruct patients to take gabapentin only as prescribed.
- Advise patient to not stop taking gabapentin without first talking to their health care provider.
- If patients break the scored 600 or 800 mg tablet in order to administer a half-tablet, they should take the unused half-tablet as the next dose. Half-tablets not used within several days of breaking the scored tablet should be discarded.
- Advise patients that if dosage is reduced, discontinued, or substituted with an alternative medication, this should be done gradually over minimum of 1 week.
- Patients should be advised that gabapentin may increase the risk of suicidal thoughts and behavior.
- Patients should be alert for the emergence or worsening of symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm and report any concerns immediately to health care providers.
- Patients should be advised that gabapentin may cause dizziness, somnolence, and other symptoms and signs of CNS depression. They should neither drive a car not operate other complex machinery until they have gained sufficient experience on gabapentin to gauge whether or not it affects their mental and/or motor performance adversely.
- Patients who require concomitant treatment with morphine nay experience increases in gabapentin concentrations. Patients should be carefully observed for signs of CNS depression, such as somnolence, and the dose of gabapentin or morphine should be reduced appropriately.
- Patients should be advised to enroll in the North American Antiepileptic Drug (NAAED) Pregnancy Registry if they become pregnant.
- Patient should be instructed that a rash or other signs or symptoms of hypersensitivity (such as fever or lymphadenopathy) may herald a serious medical event.
- The patient should report any such occurrence to a physician immediately.
- Advise patients to not drink alcohol or take other medicines that make them sleepy or dizzy while taking gabapentin without first talking to their health care provider.
Indications
Dosing
(Adult):
General Notes: Given orally with or without food. Swallow capsules whole with water.
(Pediatrics):
Renal Dosing
Dosage Forms
Warnings
Overdose
Special Populations
Breastfeeding
Chemical Structure
Mechanism of Action
Pharmacokinetics
Counseling Points
MESH Terms & Keywords
|
|---|
|





