Alogliptin (Nesina): Drug Monograph
|
|---|
- An adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
- Limitation of Use: Not for treatment of type 1 diabetes or diabetic ketoacidosis.
- Normal renal function (CrCl > 60 mL/min) or mild renal impairment is 25 mg once daily.
- CrCl ≥ 30 to < 60 mL/min = 12.5 mg once daily
- CrCl ≥ 15 to < 30 mL/min = 6.25 mg once daily
- End-stage renal disease (ESRD) or CrCl < 15 mL/min = 6.25 mg once daily. Timing of hemodialysis does not matter.
- None for mild to moderate impairment.
- Not studied in patients with severe impairment.
- History of serious hypersensitivity reaction to alogliptin-containing products, such as anaphylaxis, angioedema or severe cutaneous adverse reactions.
- Acute pancreatitis
- Hypersensitivity
- Hepatic effects - Causality cannot be excluded for hepatic failure, sometimes fatal.
- Hypoglycemia
- Macrovascular outcomes - no clinical studies establishing conclusive evidence of macrovascular risk reduction with alogliptin.
- No serious adverse events were observed at doses of 800 mg to healthy subjects and doses of 400 mg once daily for 14 days to patients with type 2 diabetes.
- In event of overdose, institute the necessary clinical monitoring and supportive therapy as dictated by the patient's clinical status.
- Hemodialysis is unlikely to be beneficial since alogliptin is minimally dialyzable.
- Alogliptin is primarily renally excreted.
- Cytochrome (CYP) P450-related metabolism is negligible. No significant drug-drug interactions were observed with the CYP-substrates or inhibitors tested or with renally excreted drugs.
- Pregnancy: Pregnancy Category B
- Labor and Delivery: None
- Nursing Mothers: It is not know whether alogliptin is excreted in human milk.
- Renal Impairment: Adjust dose if moderate or severe renal impairment or end-stage renal disease (ERSD).
- Hepatic Impairment: No dose adjustments are required in patients with mild to moderate hepatic impairment. Alogliptin has not been studied in patients with severe hepatic impairment.
- Pediatric Patients: Safety and effectiveness in pediatric patients have not been established.
- Geriatric Patients: No overall differences in safety or effectiveness were observed between patients 65 years and over and younger patients.
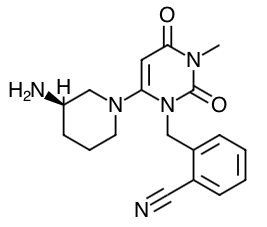
- Scientific Name: Alogliptin is prepared as a benzoate salt, which is identified as 2-({6-[(3R)-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl}methyl)benzonitrile monobenzoate.
- Empirical Formula: C18H21N5O2∙C7H6O2
- Molecular Weight: 461.51 daltons
- Upon eating, concentrations of the incretin hormones such as glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are released into the bloodstream from the small intestine to cause glucose-dependent insulin release.
- GLP-1 also lowers glucagon secretion from pancreatic alpha cells, reducing hepatic glucose production
- These hormones are inactivated by the DPP-4 enzyme within minutes. Thus, inhibitors of DDP-4 will result increase concentrations of these incretin hormones.
- Alogliptin selectively binds to and inhibits DPP-4 but not DPP-8 or DPP-9 activity in vitro at concentrations approximating therapeutic
- Onset of Action: Peak inhibition of DPP-4 occurs within 1-2 hours after dosing and remains above 90% inhibition for 24 hours when doses of 25 mg or more are used.
- In a 16-week, double-blind, placebo-controlled study, alogliptin 25 mg demonstrated decreases in postprandial glucagon while increasing postprandial active GLP-1 levels compared to placebo over an eight-hour period following a standardized meal. In this study, alogliptin 25 mg demonstrated decreases in two-hour postprandial glucose compared to placebo (-30 mg/dL versus 17 mg/dL, respectively).
- Cardiac Electrophysiology: In a randomized, placebo-controlled, four-arm, parallel-group study, 257 subjects were administered either alogliptin 50 mg, alogliptin 400 mg, moxifloxacin 400 mg or placebo once daily for a total of seven days. No increase in QTc was observed with either dose of alogliptin.
- The pharmacokinetics of alogliptin has been studied in healthy subjects and in patients with type 2 diabetes. After administration of single, oral doses up to 800 mg in healthy subjects, the peak plasma alogliptin concentration (median Tmax) occurred one to two hours after dosing. At the maximum recommended clinical dose of 25 mg, alogliptin was eliminated with a mean terminal half-life (T1/2) of approximately 21 hours. After multiple-dose administration up to 400 mg for 14 days in patients with type 2 diabetes, accumulation of alogliptin was minimal with an increase in total (i.e., AUC) and peak (i.e., Cmax) alogliptin exposures of 34% and 9%, respectively. Total and peak exposure to alogliptin increased proportionally across single doses and multiple doses of alogliptin ranging from 25 mg to 400 mg. The intersubject coefficient of variation for alogliptin AUC was 17%. The pharmacokinetics of NESINA was also shown to be similar in healthy subjects and in patients with type 2 diabetes.
- Absorption: The absolute bioavailability is ~ 100%.
- Administration of alogliptin with a high-fat meal results in no significant change in total and peak exposure to alogliptin. Alogliptin may therefore be administered with or without food.
- Volume of Distribution:417 L, indicating that the drug is well distributed into tissues.
- Protein Binding: 20% is bound to plasma proteins.
- Metabolism: Alogliptin does not undergo extensive metabolism and 60% to 71% of the dose is excreted as unchanged drug in the urine.
- Two minor metabolites were detected following administration of an oral dose of [14C] alogliptin, N-demethylated, M-I (<1% of the parent compound), and N-acetylated alogliptin, M-II (<6% of the parent compound). M-I is an active metabolite and is an inhibitor of DPP-4 similar to the parent molecule; M-II does not display any inhibitory activity toward DPP-4 or other DPP-related enzymes. In vitro data indicate that CYP2D6 and CYP3A4 contribute to the limited metabolism of alogliptin.
- Alogliptin exists predominantly as the (R)-enantiomer (>99%) and undergoes little or no chiral conversion in vivo to the (S)-enantiomer. The (S)-enantiomer is not detectable at the 25 mg dose.
- Elimination: The primary route of elimination of occurs via renal excretion (76%) with 13% recovered in the feces. The renal clearance of alogliptin (9.6 L/hr) indicates some active renal tubular secretion and systemic clearance was 14.0 L/hr.
- Renal Impairment:
- In patients with mild renal impairment (creatinine clearance [CrCl] ≥60 to <90 mL/min), an approximate 1.2-fold increase in plasma AUC of alogliptin was observed. Because increases of this magnitude are not considered clinically relevant, dose adjustment for patients with mild renal impairment is not recommended.
- In patients with moderate renal impairment (CrCl ≥30 to <60 mL/min), an approximate two-fold increase in plasma AUC of alogliptin was observed. To maintain similar systemic exposures of NESINA to those with normal renal function, the recommended dose is 12.5 mg once daily in patients with moderate renal impairment.
- In patients with severe renal impairment (CrCl ≥15 to <30 mL/min) and ESRD (CrCl <15 mL/min or requiring dialysis), an approximate three- and four-fold increase in plasma AUC of alogliptin were observed, respectively. Dialysis removed approximately 7% of the drug during a three-hour dialysis session. Alogliptin may be administered without regard to the timing of the dialysis. To maintain similar systemic exposures of alogliptin to those with normal renal function, the recommended dose is 6.25 mg once daily in patients with severe renal impairment, as well as in patients with ESRD requiring dialysis.
- Hepatic Impairment: Total exposure to alogliptin was approximately 10% lower and peak exposure was approximately 8% lower in patients with moderate hepatic impairment (Child-Pugh Grade B) compared to healthy subjects. The magnitude of these reductions is not considered to be clinically meaningful. Patients with severe hepatic impairment (Child-Pugh Grade C) have not been studied. Use caution when administering alogliptin to patients with liver disease.
- Gender: No dose adjustment of alogliptin is necessary based on gender. Gender did not have any clinically meaningful effect on the pharmacokinetics of alogliptin.
- Geriatric: No dose adjustment of alogliptin is necessary based on age. Age did not have any clinically meaningful effect on the pharmacokinetics of alogliptin.
- Pediatric: Studies characterizing the pharmacokinetics of alogliptin in pediatric patients have not been performed.
- Race: No dose adjustment of alogliptin is necessary based on race. Race (White, Black, and Asian) did not have any clinically meaningful effect on the pharmacokinetics of alogliptin.
- Drug Interactions
- In Vitro Assessment of Drug Interactions: In vitro studies indicate that alogliptin is neither an inducer of CYP1A2, CYP2B6, CYP2C9, CYP2C19 and CYP3A4, nor an inhibitor of CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP3A4 and CYP2D6 at clinically relevant concentrations.
- Tell patients to take alogliptin only as prescribed and if a dose is missed, advise patients not to double their next dose.
- Acute pancreatitis. Persistent, severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting, is the hallmark symptom of acute pancreatitis. Promptly discontinue alogliptin and contact their physician if persistent severe abdominal pain occurs.
- Allergic reactions. Symptoms of allergic reactions may include skin rash, hives and swelling of the face, lips, tongue and throat that may cause difficulty in breathing or swallowing. Discontinue alogliptin and seek medical advice promptly.
- Liver injury. If
signs or symptoms of liver injury occur, discontinue alogliptin and seek
medical advice promptly.
- Hypoglycemia can occur, particularly when an insulin secretagogue or insulin is used in combination with alogliptin. Explain the risks, symptoms and appropriate management of hypoglycemia.
Indications
Renal Dosing
Hepatic Dosing
Contraindications
Warnings
Overdose
Drug Interactions
Special Populations
Chemical Structure
Mechanism of Action
Pharmacodynamics
Pharmacokinetics
Specific Populations:
Counseling Points
MESH Terms & Keywords
|
|---|
|





