Digoxin (Lanoxin) is a well known cardiac glycoside and has been used
for many years to treat several cardiovascular problems, such as heart
failure and rate control in patients with atrial arrhythmias who
experience rapid ventricular response.1,2 As it relates to heart
failure, it is important to note that this is in the context of systolic
heart failure, not diastolic heart failure where digoxin could
potentially worsen that condition.1 This distinction is not only
important clinically, but forms the context for the question presented.
As a quick review, patients with systolic heart failure will usually
have reductions in cardiac output (CO). This reduction in CO primarily
occurs from a reduction in stroke volume (SV). Stroke volume is
influenced by a number of factors such as 1) inotropy (force of
ventricular contraction), 2) preload (volume of blood returning back to
the heart or filling the ventricles during diastole), and 3) afterload
(resistance to forward blood flow during systole). Therefore, anything
that increases any one or more of these factors will increase SV and
thus, ultimately increases CO (assuming the pulse does not change or
decrease).
Most clinicians recognize that digoxin's role in helping to control
symptoms associated with systolic heart failure has to do with its
inotropic effect or ability to increase the force of contraction.3 Some
of those clinicians will go on to say that it does this by increasing
the intracellular calcium (Ca2+) concentrations. While that is all true,
the real question that connects this thought process is how does
digoxin cause this increase in cytosolic calcium concentrations and how
does that ultimately increase inotropy? In order to understand the role
of digoxin in systolic heart failure, the clinician must understand the
normal physiology of the cardiac cycle that causes ventricular
contraction (systole) and relaxation (diastole).
What happens during normal cardiac muscle contraction?
Upon ventricular depolarization (systole), sodium (Na+) moves into the
cardiac myocyte (during phase 0 of the cardiac action potential; see
figure below). Shortly thereafter, potassium (K+) will start to move out
of the cardiac myocyte to be placed in the extracellular environment
(this is phase 1 in the action potential). During this time, the
cytosolic concentrations of Ca2+ are known to increase rapidly through a
number of mechanisms (representing phase 2 of the action potential).4
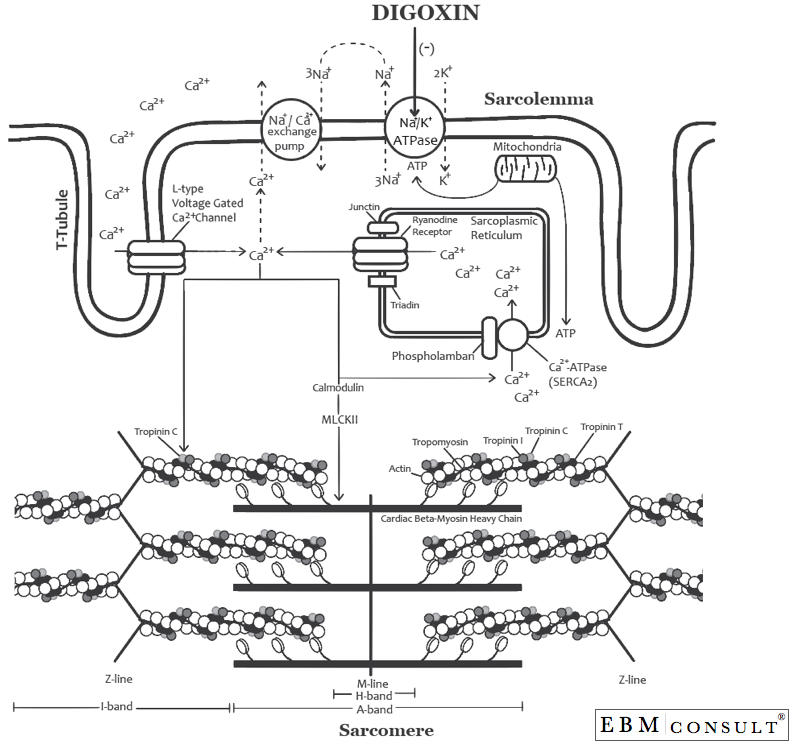
As shown in the second figure, Ca2+ comes into the cardiac myocyte via
the L-type voltage gated Ca2+ channels that line the T-tubule of the
sarcomere (this channel has also been called the DHPR = dihydropyridine
receptor and is the receptor that the "dihydropyridine" type calcium
channel blockers (i.e., diltiazem and verapamil) inhibit). Once the Ca2+
gets into the cytosol it binds to calmodulin to activate the
Ca2+/calmodulin-dependent protein kinase (also known as, myosin light
chain kinase II (MLKII or CaMKII).5 Once MLKII has been formed it can do
a lot of things, one of which is increasing cytosolic Ca2+. MLKII does
this by two mechanisms: 1) it can phosphorylate the ryanodine receptors
(RyR) on the sarcoplasmic reticulum, which causes Ca2+ to move from
inside the sarcoplasmic reticulum into the cytosol (cytoplasm) and at
the same time it can 2) phosphorylate phospholamban, which puts Ca2+
inside the sarcoplasmic reticulum via Ca2+-ATPase (SERCA2) during
repolarization in preparation for being pushed out via the RyR upon the
next cardiac depolarization.6,7 In most situations these are working
together, however, sympathetic stimulation (as seen in heart failure)
can also increase the activity of phospholamban during repolarization,
thereby putting more Ca2+ into the sarcoplasmic reticulum that is now
ready for the next repolarization or action potential. This is the
attempt by the human body to increase inotropy with sympathetic
stimulation especially during left ventricular systolic heart failure
where cardiac output is compromised.
This increase in cytosolic Ca2+ then allows Ca2+ to bind to troponin
C, which moves tropomyosin thereby allowing myosin and actin to interact
with each other to cause a contraction (or shortening of the
sarcomere).8,9 The greater the concentration of cytosolic Ca2+ the
greater this process can occur. Now upon repolarization (during
diastole) much of the process is being reversed. Calcium will then move
back into the sarcoplasmic reticulum or can be moved outside of the
cardiac myocyte via the Na+/Ca2+ exchange pump, thus allowing for
relaxation of the cardiac myocytes during diastole. Furthermore, the
Na+/K+ ATPase pump resets the membrane potential by kicking 3 Na+ ions
out of the cell and bringing back 2 K+ ions back into the cell during
repolarization (representing phase 3 of the action potential).
What happens to this physiologic process if the patient is given digoxin?
Once distributed to the heart, digoxin binds to the phosphorylated form
of the alpha subunit of the Na+/K+ ATPase pumps and inhibits their
activity (see the figure below).10,11 This causes the intracellular or
cytosolic Na+ concentration to remain higher, which in turn disrupts the
Na+ gradient needed to operate the Na+/ Ca2+ exchange pump because it
works by bringing 3 Na+ from outside of the cardiac myocyte into the
myocyte and in exchange it would take a Ca2+ from inside the myocyte and
move it outside the cell. Therefore, a greater concentration of
cytosolic Ca2+ occurs inside the cell with digoxin, thereby allowing for
a greater degree of binding to troponin C and eventually myosin/actin
binding thus allowing for a greater force of contraction (or inotropy).
What does this mean clinically (as it relates to inotropy)?
Well unfortunately not as much as it sounds or would be desired. Despite
having a unique and useful mechanism of action for patients with lower
CO, the Dig Trial failed to show a reduction in mortality in patients
with heart failure.12 However, digoxin is known to reduce symptoms and
hospitalizations associated to heart failure, which it is why it is
recommended in Stage C systolic heart failure per the AHA/ACC
guidelines.1,12 Lastly, it is important to keep in mind this small
beneficial effect is only seen with therapeutic levels. Since digoxin
has a narrow therapeutic index, appropriate monitoring of drug
concentrations is necessary, especially in patients with impaired or
changing renal function and starting new medications that are known
inhibitors of the efflux pump, P-glycoprotein.
References:
- Jessup M, Abraham WT, Casey WT et al. 2009 focused update: ACCF/AHA
Guidelines for the Diagnosis and Management of Heart Failure in Adults: a
report of the American College of Cardiology Foundation/American Heart
Association Task Force on Practice Guidelines: developed in
collaboration with the International Society for Heart and Lung
Transplantation. Circulation 2009;119:1977-2016.
- Cheng JW, Rybak I. Use of digoxin for heart failure and atrial
fibrillation in elderly patients. Am J Geriatr Pharmacother
2010;8:419-27.
- Little WC, Rossi JR, Freeman GL. Comparison of effects of
dobutamine and ouabain on left ventricular contraction and relaxation in
closed-chest dogs. J Clin Invest 1987:80;613-620.
- Zhang L, Kelley J, Schmeisser G et al. Complex formation between
junctin, triadin, calsequestrin, and the ryanodine receptor. Proteins
of the cardiac junctional sarcoplasmic reticulum membrane. J Biol Chem
1997;272:23389-97.
- Couchonnal LF, Anderson ME. The role of calmodulin kinase II in myocardial physiology and disease. Physiology 2008;23:151-9.
- Lanner JT, Georgiou DK, Joshi AD et al. Ryanodine receptors:
structure, expression, molecular details, and function in calcium
release. Cold Spring Harb Perspect Biol 2010;2:a003996.
- Beard NA, Wei L, Dulhunty AF. Control of muscle ryanodine
receptor calcium release channels by proteins in the sarcoplasmic
reticulum lumen. Clin Exp Pharmacol Physiol 2009;36:340-5.
- Kamm KE, Stull JT. Signalling to myosin regulatory light chain in sarcomeres. J Biol Chem 2011;286:9941-7.
- Ding P, Huang J, Battiprolu PK et al. Cardiac myosin light chain
kinase is necessary for myosin regulatory light chain phosphorylation
and cardiac performance in vivo. J Biol Chem 2010;285:40819-29.
- Li PW, Ho CS, Swaminathan R et al. The chronic effects of
long-term digoxin administration on Na+/K(+)-ATPase activity in rat
tissues. Int J Cardiol 1993;40:95-100.
- Eichhorn EJ, Gheorghiade M. Digoxin. Prog Cardiovasc Dis 2002;44:251-66.
- The Digitalis Investigation Group, The effect of digoxin on
mortality and morbidity in patients with heart failure, N Engl J Med
1997;336;525-533.






