Gout
has not only been a medical problem for centuries, but is increasing in
prevalence across the world.1-3 This increase in prevalence may be due to
people living longer, changes in dietary patterns that have led to increases in
obesity and development of metabolic syndrome.1,4 While not all patients
who have hyperuricemia are symptomatic, it is well known that the concentration
of serum uric acid directly correlates with the development of gout.5,6
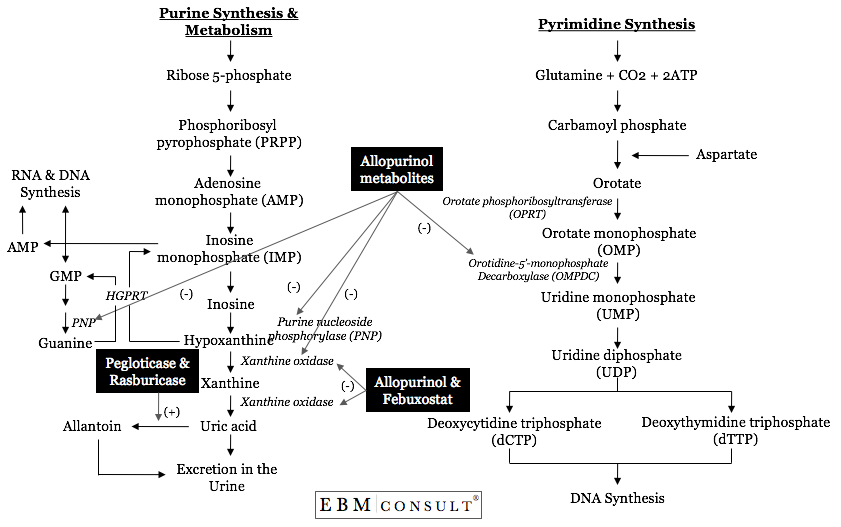
Normally, the enzyme xanthine oxidase (XO) is needed to convert the purine base
hypoxanthine to xanthine and xanthine to uric acid (see figure).5 When
the uric acid levels increase beyond 7 mg/dL in men and 6 mg/dL in
postmenopausal females, the risk for precipitation of crystals can occur.5,6
For the majority of patients, the mainstay of treatment for decreasing serum
uric acid concentrations has been with inhibitors of XO, such as allopurinol
(Zyloprim; Aloprim) and febuxostat (Uloric), along with changes in diet and
lifestyle, to achieve a target serum uric acid level of < 6 mg/dL.7,8
Do
allopurinol and febuxostat inhibit XO differently?
Yes. As stated above, hypoxanthine is a product generated in
the metabolism of purines (guanine and cytosine) and, in the presence of
functional XO, is metabolized to xanthine and again to uric acid where it then
be eliminated primarily in the urine.5 Therefore, an analogue (something
structurally similar) of purines can then compete with hypoxanthine and
xanthine for metabolism by XO. This is primarily where allopurinol and
febuxostat are different. Allopurinol and its metabolites are structural
analogues of both purines and pyrimidines and thus can affect enzymes in both
purine and pyrimidine metabolic pathways, whereas febuxostat is a non-purine
inhibitor of XO only.7-11
How
does allopurinol influence more than XO activity?
Since allopurinol is a purine analogue, it also undergoes
metabolism by XO, which results in the production to its most active metabolite
(oxypurinol).7,12 It is the oxypurinol that binds very tightly to the
reduced form of XO, resulting in a suicide inhibition of XO.12 In
addition, allopurinol is also metabolized by the enzymes hypoxanthine-guanine
phosphoribosyltransferase (HGPRT) and orotate phosphoribosyltransferase (OPRT)
that result in the production of nucleotide analogues.9-11,13
Unfortunately, these metabolites can also inhibit purine nucleoside
phosphorylase (PNP) in purine metabolism and orotidine-5'-monophosphate
decarboxylase (OMPDC), which are needed in the synthesis of pyrimidines that
will eventually be used for both RNA and DNA synthesis.10,14-16 It has
been speculated that allopurinol's influence on these other pathways may be
linked to some of allopurinol's side effects. Since febuxostat is not a
structural analogue of either purines or pyrimidines, it does not interfere
with the activity of these other pathways.8,17 As such, it is a specific
inhibitor of XO only.17 While febuxostat is a potent inhibitor of XO, it
does so by inhibiting both the reduced and oxidized forms of XO, which is
another difference from allopurinol.
Do
these pharmacologic principles translate into any differences in the reduction
of serum uric acid levels?
Several studies comparing the most commonly used doses of allopurinol at 300 mg
daily, showed the febuxostat, at mainly 80 mg, resulted in a greater percentage
of patients being able to achieve serum uric acid levels < 6 mg/dL
(allopurinol, 42% versus febuxostat, 72%).8
While
not specifically related to the mechanism of action in gout, febuxostat, unlike
allopurinol, is a substrate of uridine diphosphate glucuronosyltransferase
(UGT) 1A1, 1A3, 1A9, 2B7 and cytochrome P-450 (CYP) 1A2, 2C8, and 2C9.8
In addition, febuxostat is also known to be a weak inhibitor of CYP2D6.8 As
such, febuxostat may be subject to more drug interactions than compared to
allopurinol. However, it is imperative that clinicians recognize that
both allopurinol and febuxostat can inhibit the XO-mediated metabolism of
azathioprine (Imuran), thereby putting patients at an increased risk for
life-threatening bone marrow suppression.7,8,18 Lastly, febuxostat also
differs from allopurinol in that dose reductions are not needed in patients
with mild to moderate renal impairment, whereas allopurinol does need dose
reductions.8
References:
- Bieber JD, Terkeltaub RA. Gout: on the brink of novel therapeutic
options for an ancient disease. Arthritis Rheum 2004;50:2400-14.
- Wallace
KL, Reidel AA, Joseph-Ridge N et al. Increasing prevalence of gout and
hyperuricemia over 10 years among older adults in a managed care
population. J Rheumatol 2004;31:1582-7.
- Choi HK, Curhan G. Gout: epidemiology and lifestyle choices. Curr Opin Rheumatol 2005;17:341-5.
- Johnson
RJ, Rideout BA. Uric acid and diet-insights into the epidemic of
cardiovascular disease. N Engl J Med 2004;350:1071-3.
- Choi HK, Mount DB, Reginato AM et al. Pathogenesis of gout. Ann Intern Med 2005;143:499-516.
- Terkeltaub RA. Clinical practice. Gout. N Engl J Med 2003;349:1647-55.
- Allopurinol tablets product package insert. Watson Laboratories, inc.; Corona, CA. January 2006.
- Febuxostat (Uloric®) product package insert. Takeda Pharmaceuticals America, Inc. Deerfield, IL. February 2009.
- Reiter
S, Loffler W, Grobner W et al. Urinary oxipurinol-1-riboside excretion
and allopurinol-induced oroticaciduria. Adv Exp Med Biol 1986;195 Pt
A:453-60.
- Simmonds
HA, Reiter S, Davies PM et al. Orotidine accumulation in human
erythrocytes during allopurinol therapy: association with high urinary
oxypurinol-7-riboside concentrations in renal failure and in the
Lesch-Nyhan syndrome. Clin Sci (Lond) 1991;80:191-7.
- Krenitsky
TA, Elion GB, Strelitz RA et al. Ribonucleosides of allopurinol and
oxoallopurinol. Isolation from human urine, enzymatic synthesis, and
characterization. J Biol Chem 1967;242:2675-82.
- Massey
V, Komai H, Plamer G et al. On the mechanism of inactivation of
xanthine oxidase by allopurinol and other pyrazolo[3,4-d]pyrimidines. J
Biol Chem 1970;245:2837-44.
- Nelson DJ, Elion GB. Metabolic studies of high doses of allopurinol in humans. Adv Exp Mol Biol 1984;165 Pt A:167-70.
- Krenitsky
TA, Elion GB, Henderson AM et al. Inhibition of human purine
nucleoside phosphorylase. Studies with intact erythrocytes and the
purified enzyme. J Biol Chem 1968;243:2876-81.
- Nishida
Y, Kamatani M, Tanimoto K et al. Inhibition of purine nucleoside
phosphorylase activity and of T-cell function with
allopurinol-riboside. Agents Actions 1979;9:549-52.
- Kelley WN, Beardmore TD. Allopurinol: alteration in pyrimidine metabolism in man. Science 1970;169:388-90.
- Komoriya
K, Hoshide S, Takeda K et al. Pharmacokinetics and pharmacodynamics of
febuxostat (TMX-67), a non-purine selective inhibitor of xanthine
oxidase/xanthine dehydrogenase (NPSIXO) in patients with gout and/or
hyperuricemia. Nucleoside Nucleotides Nucleic Acids 2004;23:1119-22.
- Busti
AJ, Lehew DS, Nuzum DS et al. How does the interaction between
allopurinol (Zyloprim, Aloprim) and the immunosuppressant, azathioprine
(Imuran; Azasan) increase the risk of significant reductions in WBCs? Pw
Database.






