Tadalafil (Adcirca, Cialis): Drug Monograph
|
|---|
- Treatment of erectile dysfunction (ED) (Cialis)
- Treatment of the signs and symptoms of benign prostatic hyperplasia (BPH) (Cialis)
- Treatment of ED and the signs and symptoms of BPH (ED/BPH) (Cialis)
- Treatment of pulmonary arterial hypertension (WHO Group 1) to improve exercise ability (Adcirca)
- If used with finasteride to initiate BPH treatment, such use if recommended for up to 26 weeks.
- May be taken without regard to food. Do not split tablets; entire dose should be taken.
- Once-daily use: take at approximately the same time every day (Cialis). Dividing the dose (40 mg) over the course of the day is not recommended (Adcirca)
- Erectile Dysfunction (ED):
- As needed use: 10 mg by mouth prior to sexual activity.
- May increase to 20 mg or decrease to 5 mg, based on individual efficacy and tolerability. Maximum dosing frequency: once daily
- Once-daily use: 2.5 mg once daily. May increase to 5 mg once daily based on individual efficacy and tolerability
- Benign Prostatic Hyperplasia (BPH; +/- Erectile Dysfunction):
- 5 mg by mouth once daily
- Note: If initiated with finasteride: 5 mg by mouth once daily for ≤ 26 weeks
- Pulmonary Arterial Hypertension (PAH; WHO Group 1):
- 40 mg (two 20 mg tablets) by mouth once daily
- Adcirca: Mild (CrCl 51-80 mL/min) or Moderate (CrCl 31-50 mL/min):
- 20 mg once daily
- Increase to 40 mg once daily based on individual tolerability
- Adcirca: Severe (CrCl <30 mL/min and on hemodialysis):
- Avoid use
- Cialis: PRN use, CrCl 30-50 mL/min:
- 5 mg once daily
- Max Dose: 10 mg/48 hours
- Cialis: PRN use, CrCl <30 mL/min or hemodialysis:
- Max Dose: 5 mg/72 hours
- Cialis: once-daily use, ED, CrCl <30 mL/min or hemodialysis:
- Not recommended
- Cialis: once-daily use, BPH or ED/BPH, CrCl 30-50 mL/min:
- 2.5 mg; may increase to 5 mg based on individual response
- Cialis: once-daily use, BPH or ED/BPH, CrCl <30 mL/min or hemodialysis:
- Not recommended
- Adcirca, mild or moderate hepatic cirrhosis (Child-Pugh Class A/B):
- Consider 20 mg once daily
- Adcirca, severe hepatic cirrhosis (Child-Pugh Class C):
- Avoid use
- Cialis, PRN use, mild/moderate (Child-Pugh Class A/B):
- Use caution
- Cialis, PRN use, severe (Child-Pugh Class C):
- Not recommended
- Cialis, once-daily use, mild/moderate (Child-Pugh Class A/B):
- Use caution
- Cialis, once-daily use, severe (Child-Pugh Class C):
- Not recommended
- Patients using any form of organic nitrate. Tadalafil was shown to potentiate the hypotensive effect of nitrates
- Known serious hypersensitivity reaction to tadalafil
- Patients should not use if sex is inadvisable due to cardiovascular status
- Use with alpha-blockers, antihypertensives, or substantial amounts of alcohol (≥5 units) may lead to hypotension
- Not recommended in combination with alpha-blockers for the treatment of BPH because efficacy of the combination has not been adequately studied and because of the risk of blood pressure lowering. Caution is advised when used as a treatment for ED in men taking alpha-blockers.
- Prolonged erection - patients should seek emergency treatment if an erection lasts >4 hours. Use with caution in patients predisposed to priapism.
- Effects on the eye - patients should stop treatment and seek medical care if a sudden loss of vision occurs in one or both eyes, which could be a sign of non-arteritic anterior ischemic optic neuropathy (NAION). Should be used with caution, and only when the anticipated benefits outweigh the risks, in patients with a history of NAION. Patients with a "crowded" optic disc may also be at an increased risk of NAION.
- Sudden decrease or loss of hearing - patients should stop treatment and seek prompt medical attention
- Other urological conditions - prior to initiating treatment for BPH, consideration should be given to other urological conditions that may cause similar symptoms
- Headache
- Dyspepsia
- Back pain
- Myalgia
- Nasal congestion
- Flushing
- Pain in limb
- Not indicated for use in women.
- Standard supportive measures should be adopted as required.
- Hemodialysis contributes negligibly to tadalafil elimination.
- Nitrates, alpha-blockers, antihypertensives or alcohol - tadalafil can potentiate the hypotensive effects
- CYP3A4 inhibitors (e.g., ketoconazole, ritonavir) - increase tadalafil exposure requiring dose adjustment. Tadalafil for use as needed: no more than 10 mg every 72 hours. For once daily use: dose not to exceed 2.5 mg CYP3A4 inducers (e.g., rifampin) decrease tadalafil exposure
- Pregnancy: Pregnancy Category B
- Labor and Delivery: None
- Nursing Mothers: Not indicated for use in women.
- Renal Impairment:
- CrCl 30 to 50 mL/min: dosage adjustment may be needed.
- CrCl <30 mL/min or on hemodialysis: For use as needed: dose should not exceed 5 mg every 72 hours. Once daily use is not recommended.
- Hepatic Impairment: Mild or moderate: dosage adjustment may be needed. Severe: use is not recommended.
- Pediatric Patients: Not indicated for use in pediatric patients. Safety and efficacy in patients below the age of 18 years has not been established.
- Geriatric Patients: No overall differences in efficacy or safety observed between older (>65 and ≥75 years of age) and younger subjects (≤65 years of age). Therefore no dose adjustment is warranted based on age alone. However, a greater sensitivity to medications in some older individuals should be considered.
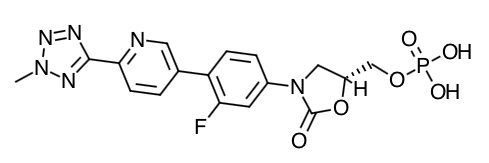
- Scientific Name: pyrazino[1′,2′:1,6]pyrido[3,4-b]indole-1,4-dione, 6-(1,3-benzodioxol-5-yl)- 2,3,6,7,12,12a-hexahydro-2-methyl-, (6R,12aR)-
- Empirical Formula: C22H19N3O4
- Molecular Weight: 389.41
- Penile erection during sexual stimulation is caused by increased penile blood flow resulting from the relaxation of penile arteries and corpus cavernosal smooth muscle. This response is mediated by the release of nitric oxide (NO) from nerve terminals and endothelial cells, which stimulates the synthesis of cGMP in smooth muscle cells. Cyclic GMP causes smooth muscle relaxation and increased blood flow into the corpus cavernosum. The inhibition of phosphodiesterase type 5 (PDE5) enhances erectile function by increasing the amount of cGMP. Tadalafil inhibits PDE5. Because sexual stimulation is required to initiate the local release of nitric oxide, the inhibition of PDE5 by tadalafil has no effect in the absence of sexual stimulation.
- The effect of PDE5 inhibition on cGMP concentration in the corpus cavernosum and pulmonary arteries is also observed in the smooth muscle of the prostate, the bladder and their vascular supply. The mechanism for reducing BPH symptoms has not been established.
- Studies in vitro have demonstrated that tadalafil is a selective inhibitor of PDE5. PDE5 is found in the smooth muscle of the corpus cavernosum, prostate, and bladder as well as in vascular and visceral smooth muscle, skeletal muscle, urethra, platelets, kidney, lung, cerebellum, heart, liver, testis, seminal vesicle, and pancreas.
- In vitro studies have shown that the effect of tadalafil is more potent on PDE5 than on other phosphodiesterases. These studies have shown that tadalafil is >10,000-fold more potent for PDE5 than for PDE1, PDE2, PDE4, and PDE7 enzymes, which are found in the heart, brain, blood vessels, liver, leukocytes, skeletal muscle, and other organs. Tadalafil is >10,000-fold more potent for PDE5 than for PDE3, an enzyme found in the heart and blood vessels. Additionally, tadalafil is 700-fold more potent for PDE5 than for PDE6, which is found in the retina and is responsible for phototransduction. Tadalafil is >9,000-fold more potent for PDE5 than for PDE8, PDE9, and PDE10. Tadalafil is 14-fold more potent for PDE5 than for PDE11A1 and 40-fold more potent for PDE5 than for PDE11A4, two of the four known forms of PDE11. PDE11 is an enzyme found in human prostate, testes, skeletal muscle and in other tissues (e.g., adrenal cortex). In vitro, tadalafil inhibits human recombinant PDE11A1 and, to a lesser degree, PDE11A4 activities at concentrations within the therapeutic range. The physiological role and clinical consequence of PDE11 inhibition in humans have not been defined.
- Effects on Blood Pressure: Tadalafil 20 mg administered to healthy male subjects produced no significant difference compared to placebo in supine systolic and diastolic blood pressure (difference in the mean maximal decrease of 1.6/0.8 mm Hg, respectively) and in standing systolic and diastolic blood pressure (difference in the mean maximal decrease of 0.2/4.6 mm Hg, respectively). In addition, there was no significant effect on heart rate.
- Effects on Blood Pressure When Administered with Nitrates: In clinical pharmacology studies, tadalafil (5 to 20 mg) was shown to potentiate the hypotensive effect of nitrates. Therefore, the use of tadalafil in patients taking any form of nitrates is contraindicated. A study was conducted to assess the degree of interaction between nitroglycerin and tadalafil, should nitroglycerin be required in an emergency situation after tadalafil was taken. This was a double-blind, placebo-controlled, crossover study in 150 male subjects at least 40 years of age (including subjects with diabetes mellitus and/or controlled hypertension) and receiving daily doses of tadalafil 20 mg or matching placebo for 7 days. Subjects were administered a single dose of 0.4 mg sublingual nitroglycerin (NTG) at pre-specified timepoints, following their last dose of tadalafil (2, 4, 8, 24, 48, 72, and 96 hours after tadalafil). The objective of the study was to determine when, after tadalafil dosing, no apparent blood pressure interaction was observed. In this study, a significant interaction between tadalafil and NTG was observed at each timepoint up to and including 24 hours. At 48 hours, by most hemodynamic measures, the interaction between tadalafil and NTG was not observed, although a few more tadalafil subjects compared to placebo experienced greater blood pressure lowering at this timepoint. After 48 hours, the interaction was not detectable.Therefore, administration with nitrates is contraindicated. In a patient who has taken tadalafil, where nitrate administration is deemed medically necessary in a life-threatening situation, at least 48 hours should elapse after the last dose of tadalafil before nitrate administration is considered. In such circumstances, nitrates should still only be administered under close medical supervision with appropriate hemodynamic monitoring.
- Effects on Blood Pressure When Administered With Alpha-Blockers: Six randomized, double-blinded, crossover clinical pharmacology studies were conducted to investigate the potential interaction of tadalafil with alpha-blocker agents in healthy male subjects. In four studies, a single oral dose of tadalafil was administered to healthy male subjects taking daily (at least 7 days duration) an oral alpha-blocker. In two studies, a daily oral alpha-blocker (at least 7 days duration) was administered to healthy male subjects taking repeated daily doses of tadalafil.
- Doxazosin — Three clinical pharmacology studies were conducted with tadalafil and doxazosin, an alpha[1]- adrenergic blocker.
- In the first doxazosin study, a single oral dose of tadalafil 20 mg or placebo was administered in a 2-period, crossover design to healthy subjects taking oral doxazosin 8 mg daily (N=18 subjects). Doxazosin was administered at the same time as tadalafil or placebo after a minimum of seven days of doxazosin dosing. Blood pressure was measured manually at 1, 2, 3, 4, 5, 6, 7, 8, 10, 12, and 24 hours after tadalafil or placebo administration. Outliers were defined as subjects with a standing systolic blood pressure of <85 mm Hg or a decrease from baseline in standing systolic blood pressure of >30 mm Hg at one or more time points. There were nine and three outliers following administration of tadalafil 20 mg and placebo, respectively. Five and two subjects were outliers due to a decrease from baseline in standing systolic BP of >30 mm Hg, while five and one subject were outliers due to standing systolic BP <85 mm Hg following tadalafil and placebo, respectively. Severe adverse events potentially related to blood-pressure effects were assessed. No such events were reported following placebo. Two such events were reported following administration of tadalafil. Vertigo was reported in one subject that began 7 hours after dosing and lasted about 5 days. This subject previously experienced a mild episode of vertigo on doxazosin and placebo. Dizziness was reported in another subject that began 25 minutes after dosing and lasted 1 day. No syncope was reported.
- In the second doxazosin study, a single oral dose of tadalafil 20 mg was administered to healthy subjects taking oral doxazosin, either 4 or 8 mg daily. The study (N=72 subjects) was conducted in three parts, each a 3-period crossover. In part A (N=24), subjects were titrated to doxazosin 4 mg administered daily at 8 a.m. Tadalafil was administered at either 8 a.m., 4 p.m., or 8 p.m. There was no placebo control. In part B (N=24), subjects were titrated to doxazosin 4 mg administered daily at 8 p.m. Tadalafil was administered at either 8 a.m., 4 p.m., or 8 p.m. There was no placebo control. In part C (N=24), subjects were titrated to doxazosin 8 mg administered daily at 8 a.m. In this part, tadalafil or placebo were administered at either 8 a.m. or 8 p.m. Blood pressure was measured by ABPM every 15 to 30 minutes for up to 36 hours after tadalafil or placebo. Subjects were categorized as outliers if one or more systolic blood pressure readings of <85 mm Hg were recorded or one or more decreases in systolic blood pressure of >30 mm Hg from a time-matched baseline occurred during the analysis interval. Of the 24 subjects in part C, 16 subjects were categorized as outliers following administration of tadalafil and 6 subjects were categorized as outliers following placebo during the 24-hour period after 8 a.m. dosing of tadalafil or placebo. Of these, 5 and 2 were outliers due to systolic BP <85 mm Hg, while 15 and 4 were outliers due to a decrease from baseline in systolic BP of >30 mm Hg following tadalafil and placebo, respectively. During the 24-hour period after 8 p.m. dosing, 17 subjects were categorized as outliers following administration of tadalafil and 7 subjects following placebo. Of these, 10 and 2 subjects were outliers due to systolic BP <85 mm Hg, while 15 and 5 subjects were outliers due to a decrease from baseline in systolic BP of >30 mm Hg, following tadalafil and placebo, respectively. Some additional subjects in both the tadalafil and placebo groups were categorized as outliers in the period beyond 24 hours. Severe adverse events potentially related to blood-pressure effects were assessed. In the study (N=72 subjects), 2 such events were reported following administration of tadalafil (symptomatic hypotension in one subject that began 10 hours after dosing and lasted approximately 1 hour, and dizziness in another subject that began 11 hours after dosing and lasted 2 minutes). No such events were reported following placebo. In the period prior to tadalafil dosing, one severe event (dizziness) was reported in a subject during the doxazosin run-in phase.
- In the third doxazosin study, healthy subjects (N=45 treated; 37 completed) received 28 days of once per day dosing of tadalafil 5 mg or placebo in a two-period crossover design. After 7 days, doxazosin was initiated at 1 mg and titrated up to 4 mg daily over the last 21 days of each period (7 days on 1 mg; 7 days of 2 mg; 7 days of 4 mg doxazosin). Blood pressure was measured manually pre-dose at two time points (-30 and -15 minutes) and then at 1, 2, 3, 4, 5, 6, 7, 8, 10, 12 and 24 hours post dose on the first day of each doxazosin dose, (1 mg, 2 mg, 4 mg), as well as on the seventh day of 4 mg doxazosin administration. Following the first dose of doxazosin 1 mg, there were no outliers on tadalafil 5 mg and one outlier on placebo due to a decrease from baseline in standing systolic BP of >30 mm Hg. There were 2 outliers on tadalafil 5 mg and none on placebo following the first dose of doxazosin 2 mg due to a decrease from baseline in standing systolic BP of >30 mm Hg. There were no outliers on tadalafil 5 mg and two on placebo following the first dose of doxazosin 4 mg due to a decrease from baseline in standing systolic BP of >30 mm Hg. There was one outlier on tadalafil 5 mg and three on placebo following the first dose of doxazosin 4 mg due to standing systolic BP <85 mm Hg. Following the seventh day of doxazosin 4 mg, there were no outliers on tadalafil 5 mg, one subject on placebo had a decrease >30 mm Hg in standing systolic blood pressure, and one subject on placebo had standing systolic blood pressure <85 mm Hg. All adverse events potentially related to blood pressure effects were rated as mild or moderate. There were two episodes of syncope in this study, one subject following a dose of tadalafil 5 mg alone, and another subject following coadministration of tadalafil 5 mg and doxazosin 4 mg.
- Tamsulosin
- In the first tamsulosin study, a single oral dose of tadalafil 10, 20 mg, or placebo was administered in a 3 period, crossover design to healthy subjects taking 0.4 mg once per day tamsulosin, a selective alpha[1A]- adrenergic blocker (N=18 subjects). Tadalafil or placebo was administered 2 hours after tamsulosin following a minimum of seven days of tamsulosin dosing. Blood pressure was measured manually at 1, 2, 3, 4, 5, 6, 7, 8, 10, 12, and 24 hours after tadalafil or placebo dosing. There were 2, 2, and 1 outliers (subjects with a decrease from baseline in standing systolic blood pressure of >30 mm Hg at one or more time points) following administration of tadalafil 10 mg, 20 mg, and placebo, respectively. There were no subjects with a standing systolic blood pressure <85 mm Hg. No severe adverse events potentially related to blood-pressure effects were reported. No syncope was reported.
- In the second tamsulosin study, healthy subjects (N=39 treated; and 35 completed) received 14 days of once per day dosing of tadalafil 5 mg or placebo in a two-period crossover design. Daily dosing of tamsulosin 0.4 mg was added for the last seven days of each period. Blood pressure was measured manually pre-dose at two time points (-30 and -15 minutes) and then at 1, 2, 3, 4, 5, 6, 7, 8, 10, 12, and 24 hours post dose on the first, sixth and seventh days of tamsulosin administration. There were no outliers (subjects with a decrease from baseline in standing systolic blood pressure of >30 mm Hg at one or more time points). One subject on placebo plus tamsulosin (Day 7) and one subject on tadalafil plus tamsulosin (Day 6) had standing systolic blood pressure <85 mm Hg. No severe adverse events potentially related to blood pressure were reported. No syncope was reported.
- Alfuzosin
- A single oral dose of tadalafil 20 mg or placebo was administered in a 2-period, crossover design to healthy subjects taking once-daily alfuzosin HCl 10 mg extended-release tablets, an alpha[1]-adrenergic blocker (N=17 completed subjects). Tadalafil or placebo was administered 4 hours after alfuzosin following a minimum of seven days of alfuzosin dosing. Blood pressure was measured manually at 1, 2, 3, 4, 6, 8, 10, 20, and 24 hours after tadalafil or placebo dosing. There was 1 outlier (subject with a standing systolic blood pressure <85 mm Hg) following administration of tadalafil 20 mg. There were no subjects with a decrease from baseline in standing systolic blood pressure of >30 mm Hg at one or more time points. No severe adverse events potentially related to blood pressure effects were reported. No syncope was reported.
- Effects on Blood Pressure When Administered with Antihypertensives:
- Amlodipine: A study was conducted to assess the interaction of amlodipine (5 mg daily) and tadalafil 10 mg. There was no effect of tadalafil on amlodipine blood levels and no effect of amlodipine on tadalafil blood levels. The mean reduction in supine systolic/diastolic blood pressure due to tadalafil 10 mg in subjects taking amlodipine was 3/2 mm Hg, compared to placebo. In a similar study using tadalafil 20 mg, there were no clinically significant differences between tadalafil and placebo in subjects taking amlodipine.
- Angiotensin II receptor blockers (with and without other antihypertensives): A study was conducted to assess the interaction of angiotensin II receptor blockers and tadalafil 20 mg. Subjects in the study were taking any marketed angiotensin II receptor blocker, either alone, as a component of a combination product, or as part of a multiple antihypertensive regimen. Following dosing, ambulatory measurements of blood pressure revealed differences between tadalafil and placebo of 8/4 mm Hg in systolic/diastolic blood pressure.
- Bendrofluazide: A study was conducted to assess the interaction of bendrofluazide (2.5 mg daily) and tadalafil 10 mg. Following dosing, the mean reduction in supine systolic/diastolic blood pressure due to tadalafil 10 mg in subjects taking bendrofluazide was 6/4 mm Hg, compared to placebo.
- Enalapril: A study was conducted to assess the interaction of enalapril (10 to 20 mg daily) and tadalafil 10 mg. Following dosing, the mean reduction in supine systolic/diastolic blood pressure due to tadalafil 10 mg in subjects taking enalapril was 4/1 mm Hg, compared to placebo.
- Metoprolol: A study was conducted to assess the interaction of sustained-release metoprolol (25 to 200 mg daily) and tadalafil 10 mg. Following dosing, the mean reduction in supine systolic/diastolic blood pressure due to tadalafil 10 mg in subjects taking metoprolol was 5/3 mm Hg, compared to placebo.
- Effects on Blood Pressure When Administered with Alcohol: Alcohol and PDE5 inhibitors, including tadalafil, are mild systemic vasodilators. The interaction of tadalafil with alcohol was evaluated in 3 clinical pharmacology studies. In 2 of these, alcohol was administered at a dose of 0.7 g/kg, which is equivalent to approximately 6 ounces of 80-proof vodka in an 80-kg male, and tadalafil was administered at a dose of 10 mg in one study and 20 mg in another. In both these studies, all patients imbibed the entire alcohol dose within 10 minutes of starting. In one of these two studies, blood alcohol levels of 0.08% were confirmed. In these two studies, more patients had clinically significant decreases in blood pressure on the combination of tadalafil and alcohol as compared to alcohol alone. Some subjects reported postural dizziness, and orthostatic hypotension was observed in some subjects. When tadalafil 20 mg was administered with a lower dose of alcohol (0.6 g/kg, which is equivalent to approximately 4 ounces of 80-proof vodka, administered in less than 10 minutes), orthostatic hypotension was not observed, dizziness occurred with similar frequency to alcohol alone, and the hypotensive effects of alcohol were not potentiated. Tadalafil did not affect alcohol plasma concentrations and alcohol did not affect tadalafil plasma concentrations.
- Effects on Exercise Stress Testing: The effects of tadalafil on cardiac function, hemodynamics, and exercise tolerance were investigated in a single clinical pharmacology study. In this blinded crossover trial, 23 subjects with stable coronary artery disease and evidence of exercise-induced cardiac ischemia were enrolled. The primary endpoint was time to cardiac ischemia. The mean difference in total exercise time was 3 seconds (tadalafil 10 mg minus placebo), which represented no clinically meaningful difference. Further statistical analysis demonstrated that tadalafil was non-inferior to placebo with respect to time to ischemia. Of note, in this study, in some subjects who received tadalafil followed by sublingual nitroglycerin in the post-exercise period, clinically significant reductions in blood pressure were observed, consistent with the augmentation by tadalafil of the blood-pressure-lowering effects of nitrates.
- Effects on Vision: Single oral doses of phosphodiesterase inhibitors have demonstrated transient dose-related impairment of color discrimination (blue/green), using the Farnsworth-Munsell 100-hue test, with peak effects near the time of peak plasma levels. This finding is consistent with the inhibition of PDE6, which is involved in phototransduction in the retina. In a study to assess the effects of a single dose of tadalafil 40 mg on vision (N=59), no effects were observed on visual acuity, intraocular pressure, or pupilometry. Across all clinical studies with CIALIS, reports of changes in color vision were rare (<0.1% of patients).
- Effects on Sperm Characteristics: Three studies were conducted in men to assess the potential effect on sperm characteristics of tadalafil 10 mg (one 6 month study) and 20 mg (one 6 month and one 9 month study) administered daily. There were no adverse effects on sperm morphology or sperm motility in any of the three studies. In the study of 10 mg tadalafil for 6 months and the study of 20 mg tadalafil for 9 months, results showed a decrease in mean sperm concentrations relative to placebo, although these differences were not clinically meaningful. This effect was not seen in the study of 20 mg tadalafil taken for 6 months. In addition there was no adverse effect on mean concentrations of reproductive hormones, testosterone, luteinizing hormone or follicle stimulating hormone with either 10 or 20 mg of tadalafil compared to placebo.
- Effects on Cardiac Electrophysiology: The effect of a single 100-mg dose of tadalafil on the QT interval was evaluated at the time of peak tadalafil concentration in a randomized, double-blinded, placebo, and active (intravenous ibutilide) -controlled crossover study in 90 healthy males aged 18 to 53 years. The mean change in QTc (Fridericia QT correction) for tadalafil, relative to placebo, was 3.5 milliseconds (two-sided 90% CI=1.9, 5.1). The mean change in QTc (Individual QT correction) for tadalafil, relative to placebo, was 2.8 milliseconds (two-sided 90% CI=1.2, 4.4). A 100-mg dose of tadalafil (5 times the highest recommended dose) was chosen because this dose yields exposures covering those observed upon coadministration of tadalafil with potent CYP3A4 inhibitors or those observed in renal impairment. In this study, the mean increase in heart rate associated with a 100-mg dose of tadalafil compared to placebo was 3.1 beats per minute.
- Over a dose range of 2.5 to 20 mg, tadalafil exposure (AUC) increases proportionally with dose in healthy subjects. Steady-state plasma concentrations are attained within 5 days of once per day dosing and exposure is approximately 1.6-fold greater than after a single dose.
- Absorption: After single oral-dose administration, the maximum observed plasma concentration (Cmax) of tadalafil is achieved between 30 minutes and 6 hours (median time of 2 hours). Absolute bioavailability of tadalafil following oral dosing has not been determined. The rate and extent of absorption of tadalafil are not influenced by food; thus it may be taken with or without food.
- Distribution: The mean apparent volume of distribution following oral administration is approximately 63 L, indicating that tadalafil is distributed into tissues. At therapeutic concentrations, 94% of tadalafil in plasma is bound to proteins. Less than 0.0005% of the administered dose appeared in the semen of healthy subjects.
- Metabolism: Tadalafil is predominantly metabolized by CYP3A4 to a catechol metabolite. The catechol metabolite undergoes extensive methylation and glucuronidation to form the methylcatechol and methylcatechol glucuronide conjugate, respectively. The major circulating metabolite is the methylcatechol glucuronide. Methylcatechol concentrations are less than 10% of glucuronide concentrations. In vitro data suggests that metabolites are not expected to be pharmacologically active at observed metabolite concentrations.
- Elimination: The mean oral clearance for tadalafil is 2.5 L/hr and the mean terminal half-life is 17.5 hours in healthy subjects. Tadalafil is excreted predominantly as metabolites, mainly in the feces (approximately 61% of the dose) and to a lesser extent in the urine (approximately 36% of the dose).
- Special Populations
- Geriatric: Healthy male elderly subjects (65 years or over) had a lower oral clearance of tadalafil, resulting in 25% higher exposure (AUC) with no effect on Cmax relative to that observed in healthy subjects 19 to 45 years of age. No dose adjustment is warranted based on age alone. However, greater sensitivity to medications in some older individuals should be considered.
- Pediatric: Tadalafil has not been evaluated in individuals less than 18 years old.
- Patients with diabetes mellitus: In male patients with diabetes mellitus after a 10 mg tadalafil dose, exposure (AUC) was reduced approximately 19% and Cmax was 5% lower than that observed in healthy subjects. No dose adjustment is warranted.
- Patients with BPH: In patients with BPH following single and multiple-doses of 20 mg tadalafil, no statistically significant differences in exposure (AUC and Cmax) were observed between elderly (70 to 85 years) and younger (≤60 years of age) subjects. No dose adjustment is warranted.
- Physicians should discuss with patients the contraindication of tadalafil with regular and/or intermittent use of organic nitrates. Patients should be counseled that concomitant use with nitrates could cause blood pressure to suddenly drop to an unsafe level, resulting in dizziness, syncope, or even heart attack or stroke.
- Physicians should discuss with patients the appropriate action in the event that they experience angina chest pain requiring nitroglycerin following intake of tadalafil. In such a patient, who has taken tadalafil, where nitrate administration is deemed medically necessary for a life-threatening situation, at least 48 hours should have elapsed after the last dose of tadalafil before nitrate administration is considered. In such circumstances, nitrates should still only be administered under close medical supervision with appropriate hemodynamic monitoring. Therefore, patients who experience angina chest pain after taking tadalafil should seek immediate medical attention.
- Physicians should consider the potential cardiac risk of sexual activity in patients with preexisting cardiovascular disease. Physicians should advise patients who experience symptoms upon initiation of sexual activity to refrain from further sexual activity and seek immediate medical attention.
- Physicians should discuss with patients the potential for tadalafil to augment the blood-pressure-lowering effect of alpha-blockers and antihypertensive medications.
- Physicians should discuss with patients the clinical implications of continuous exposure to tadalafil when prescribing it for once daily use, especially the potential for interactions with medication s(e.g., nitrates, alpha-blockers, antihypertensives and potent inhibitors of cytochrome P450 3A4) and with substantial consumption of alcohol.
- There have been rare reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds. Priapism, if not treated promptly, can result in irreversible damage to the erectile tissue. Physicians should advise patients who have an erection lasting greater than 4 hours, whether painful or not, to seek emergency medical attention.
- Physicians should advise patients to stop use of all PDE5 inhibitors, including tadalafil, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision, including possible permanent loss of vision that has been reported rarely post-marketing in temporal association with the use of all PDE5 inhibitors. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors.
- Physicians should discuss with patients the increased risk of NAION in individuals who have already experienced NAION in one eye. Physicians should also discuss with patients the increased risk of NAION among the general population in patients with a "crowded" optic disc, although evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including tadalafil, for this uncommon condition.
- Physicians should advise patients to stop taking PDE5 inhibitors, including tadalafil, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including tadalafil. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors.
- Patients should be made aware that both alcohol and tadalafil, a PDE5 inhibitor, act as mild vasodilators. When mild vasodilators are taken in combination, blood-pressure-lowering effects of each individual compound may be increased. Therefore, physicians should inform patients that substantial consumption of alcohol (e.g., 5 units or greater) in combination with tadalafil can increase the potential for orthostatic signs and symptoms, including increase in heart rate, decrease in standing blood pressure, dizziness, and headache.
- The use of tadalafil offers no protection against sexually transmitted diseases. Counseling of patients about the protective measures necessary to guard against sexually transmitted diseases, including Human Immunodeficiency Virus (HIV) should be considered.
- Physicians should instruct patients on the appropriate administration of tadalafil to allow optimal use.
- For tadalafil for use as needed in men with ED, patients should be instructed to take on tablet at least 30 minutes before anticipated sexual activity. In most patients, the ability to have sexual intercourse is improved for up to 36 hours.
- For tadalafil for once daily use in men with ED or ED/BPH, patients should be instructed to take one tablet at approximately the same time every day without regard for the timing of sexual activity. Tadalafil is effective at improving erectile function over the course of therapy.
- For tadalafil for once daily use in men with BPH, patients should be instructed to take one tablet at approximately the same time every day.
Indications
Dosing (Adult)
General Dosing & Administration Notes:
Renal Dosing
Hepatic Dosing
Contraindications
Warnings
Adverse Reactions
Overdose
Drug Interactions
Special Populations
Chemical Structure
Mechanism of Action
Pharmacodynamics
Pharmacokinetics
Counseling Points
MESH Terms & Keywords
|
|---|
|

