Tacrolimus (Prograf): Drug Monograph
|
|---|
- Prophylaxis of organ rejection in patients receiving allogeneic kidney, liver, or heart transplants with concomitant adrenal corticosteroids.
- Note (Important): In heart and kidney transplant patients, azathioprine or mycophenolate mofetil coadministration is recommended
- Capsules: Take consistently, either with or without food.
- Injection should be used only as a continuous IV infusion and when the patient cannot tolerate oral administration of capsules.
- IV: Dilute product with 0.9% NaCl injection or D5 injection to concentration between 0.004 mg/mL and 0.02 mg/mL prior to use. Diluted infusion solution should be stored in glass or polyethylene containers and should be discarded after 24 hours; do not store in PVC container due to decreased stability and potential for extraction of phthalates; in situations where more dilute solution are utilized (e.g., pediatric dosing), PVC-free tubing should likewise be used to minimize the potential for significant drug absorption onto tubing. Should not be mixed or co-infused with solution of pH ≥9 (e.g., ganciclovir, acyclovir)
- Cardiac transplant:
- Capsules: 0.075 mg/kg/day by mouth as 2 divided doses, every 12 hours; administer no sooner than 6 hours after heart transplant
- IV: 0.01 mg/kg/day as a continuous infusion. If receiving tacrolimus IV infusion, give 1st oral dose 8-12 hours after discontinuing the IV infusion
- Maintenance: Lower dosages than the initial dosage may be sufficient
- Drug Level Monitoring:
- Months 1 - 3 = 10-20 ng/mL.
- Months ≥ 4 = 5-15 ng/mL
- Hepatic transplant:
- Capsules; Immediate Release Formulation: 0.1 - 0.15 mg/kg/day by mouth as 2 divided doses, every 12 hours; administer no sooner than 6 hours after liver transplant
- IV: 0.03-0.05 mg/kg/day. If receiving tacrolimus IV infusion, give 1st oral dose 8-12 hours after discontinuing the IV infusion
- Maintenance: Lower dosages than the initial dosage may be sufficient
- Drug Level Monitoring:
- Months 1 - 12 = 5-20 ng/mL
- Renal transplant:
- Capsules; Immediate Release: 0.2 mg/kg/day by mouth in combination with azathioprine or 0.1 mg/kg/day in combination with MMF/Interleukin (IL)-2 receptor antagonist; give by mouth as 2 divided doses, every 12 hours.
- May administer within 24 hours of kidney transplant, but should be delayed until renal function has recovered
- Titrate based on clinical assessments of rejection and tolerability
- Drug Level Monitoring:
- Observed tacrolimus whole blood trough concentrations with kidney transplant with azathioprine: Months 1-3: 7-20 ng/mL. Months 4-12: 5-15 ng/mL
- Observed tacrolimus whole blood trough concentrations with kidney transplant with MMF/IL-2 receptor antagonist: Months 1-12: 4-11 ng/mL
- IV: 0.03-0.05 mg/kg/day as a continuous infusion.
- Note: If receiving tacrolimus IV infusion, give 1st oral dose 8-12 hours after discontinuing the IV infusion
- Hepatic transplant:
- Capsules: 0.15-0.2 mg/kg/day as 2 divided doses, every 12 hours; administer no sooner than 6 hours after liver transplant
- Observed tacrolimus whole blood trough concentrations with liver transplant: Months 1-12: 5-20 ng/mL
- IV: 0.03-0.05 mg/kg/day
-
Liver/heart transplant, preexisting impairment:
-
Start at lower end of dosing range
-
Kidney transplant, postoperative oliguria:
-
Initial dose should be administered no sooner than 6 hours and within 24 hours of transplantation, but may be delayed until renal function shows evidence of recovery
- Lymphoma and other malignancies - risk of lymphomas, including post transplant lymphoproliferative disorder (PLTD); appears related to intensity and duration of use. Avoid prolonged exposure to UV light and sunlight
- Increased risk of bacterial, viral, fungal and protozoal infections, including opportunistic infections. Combination immunosuppression should be used with caution
- Serious, sometimes fatal outcomes, including Polyoma virus-associated nephropathy (PVAN), mostly due to BK virus, and JC virus-associated progressive multifocal leukoencephalopathy (PML); consider reducing immunosuppression
- Increased risk of CMV viremia and disease; consider reducing immunosuppression
- New onset diabetes after transplant - monitor blood glucose
- Nephrotoxicity - acute and /or chronic; reduce the dose; use caution with other nephrotoxic drugs
- Neurotoxicity - risk of posterior reversible encephalopathy syndrome, monitor for neurologic abnormalities; reduce or discontinue tacrolimus and other immunosuppressants
- Hyperkalemia - monitor serum potassium levels. Careful consideration should be given prior to use of other agents also associated with hyperkalemia
- Hypertension - may require antihypertensive therapy. Monitor relevant drug-drug interactions
- Anaphylactic reactions with IV formulations - observe patients receiving tacrolimus injection for signs and symptoms of anaphylaxis
- Use with sirolimus - not recommended in liver and heart transplant due to increased risk of serious adverse reactions
- May prolong the QT/QTc prolongation and cause Torsade de Pointes - avoid in patients with congenital long QT syndrome
- Myocardial hypertrophy - consider dosage reduction or discontinuation
- Immunizations - use of live vaccines should be avoided
- Pure red cell aplasia - discontinuation should be considered
- Gastrointestinal perforation
- Kidney transplant:
- Infection
- Tremor
- Hypertension
- Abnormal renal function
- Constipation
- Diarrhea
- Headache
- Abdominal pain
- Insomnia
- Nausea
- Liver transplant:
- Tremor
- Headache
- Diarrhea
- Hypertension
- Nausea
- Abnormal real function
- Abdominal pain
- Insomnia
- Paresthesia
- Anemia
- Pain
- Fever
- Asthenia
- Hyperkalemia
- Hypomagnesemia
- Hyperglycemia
- Heart transplant:
- Abnormal renal function
- Hypertension
- Diabetes mellitus
- CMV infection
- Tremor
- Hyperglycemia
- Leukopenia
- Infection
- Anemia
- Bronchitis
- Pericardial effusion
- Urinary tract infection
- Hyperlipemia
- Limited experience is available
- Signs and symptoms may include tremors, abnormal renal function, hypertension, and peripheral edema
- Drug is not dialyzable to any significant extent; no experience with charcoal hemoperfusion
- General supportive measures and treatment of specific symptoms should be followed
- Mycophenolic acid products - can increase MPA exposure after crossover from cyclosporine to tacrolimus; monitor for MPA-related adverse reactions and adjust MMF or MPA-dose as needed
- Nelfinavir and grapefruit juice - increased tacrolimus concentrations via CYP3A inhibition; avoid concomitant use
- CYP3A inhibitors - increased tacrolimus concentrations; monitor concentrations and adjust tacrolimus dose as needed with concomitant use
- CYP3A4 inducers - decreased tacrolimus concentrations; monitor concentrations and adjust tacrolimus dose as needed with concomitant use
- Pregnancy: Pregnancy Category C
- Labor and Delivery: None
- Nursing Mothers: Tacrolimus is excreted in human milk. Patients should discontinue nursing taking into consideration the importance of the drug to the mother
- Renal Impairment: Administer at the lower end of the recommended starting dose. Monitor renal function in patients with impaired function
- Hepatic Impairment: Administer at the lower end of the recommended starting dose
- Pediatric Patients: Safety and efficacy in pediatric kidney and heart transplant patients have not been established. Successful liver transplants have been performed in patients (ages up to 16 years) using tacrolimus. Pediatric patients generally require higher doses to maintain blood trough concentrations of tacrolimus similar to adult patients
- Geriatric Patients: Dose selection should be cautious, usually starting at the low end of the dosing range
- Tacrolimus is excreted in human milk. Patients should discontinue nursing taking into consideration the importance of the drug to the mother
-
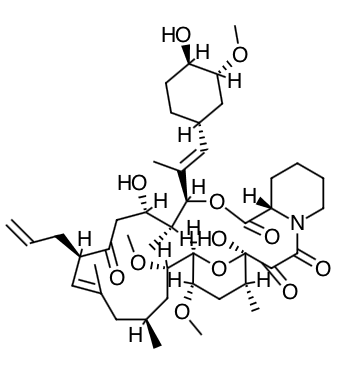
Scientific Name: [3S- [3R*[E(1S*,3S*,4S*)], 4S*,5R*,8S*,9E,12R*,14R*,15S*,16R*,18S*,19S*,26aR*]] - 5,6,8,11,12,13,14,15,16,17,18,19,24,25,26,26a-hexadecahydro-5,19-dihydroxy-3-[2-(4-hydroxy-3-methoxycyclohexyl)-1- methylethenyl]-14,16-dimethoxy-4,10,12,18-tetramethyl-8-(2-propenyl)-15,19-epoxy-3H-pyrido[2,1-c][1,4] oxaazacyclotricosine-1,7,20,21(4H,23H)-tetrone, monohydrate
-
Empirical Formula: C44H69NO12ŸH2O
-
Molecular Weight: 822.03
- Tacrolimus inhibits T-lymphocyte activation, although the exact mechanism of action is not known. Experimental evidence suggests that tacrolimus binds to an intracellular protein, FKBP-12. A complex of tacrolimus-FKBP-12, calcium, calmodulin, and calcineurin is then formed and the phosphatase activity of calcineurin inhibited. This effect may prevent the dephosphorylation and translocation of nuclear factor of activated T-cells (NF-AT), a nuclear component thought to initiate gene transcription for the formation of lymphokines (such as interleukin-2, gamma interferon). The net result is the inhibition of T-lymphocyte activation (i.e., immunosuppression).
- Tacrolimus prolongs the survival of the host and transplanted graft in animal transplant models of liver, kidney, heart, bone marrow, small bowel and pancreas, lung and trachea, skin, cornea, and limb.
- In animals, tacrolimus has been demonstrated to suppress some humoral immunity and, to a greater extent, cell-mediated reactions such as allograft rejection, delayed type hypersensitivity, collagen-induced arthritis, experimental allergic encephalomyelitis, and graft versus host disease.
- Absorption:
- Absorption of tacrolimus from the gastrointestinal tract after oral administration is incomplete and variable.
- The absolute bioavailability of tacrolimus was 17±10% in adult kidney transplant patients (N=26), 22±6% in adult liver transplant patients (N=17), 23±9% in adult heart transplant patients (N=11) and 18±5% in healthy volunteers (N=16).
- A single dose trial conducted in 32 healthy volunteers established the bioequivalence of the 1 mg and 5 mg capsules. Another single dose trial in 32 healthy volunteers established the bioequivalence of the 0.5 mg and 1 mg capsules. Tacrolimus maximum blood concentrations (Cmax) and area under the curve (AUC) appeared to increase in a dose- proportional fashion in 18 fasted healthy volunteers receiving a single oral dose of 3, 7, and 10 mg. In 18 kidney transplant patients, tacrolimus trough concentrations from 3 to 30 ng/mL measured at 10-12 hours post-dose (Cmin) correlated well with the AUC (correlation coefficient 0.93). In 24 liver transplant patients over a concentration range of 10 to 60 ng/mL, the correlation coefficient was 0.94. In 25 heart transplant patients over a concentration range of 2 to 24 ng/mL, the correlation coefficient was 0.89 after an oral dose of 0.075 or 0.15 mg/kg/day at steady-state. Food Effects: The rate and extent of tacrolimus absorption were greatest under fasted conditions. The presence and composition of food decreased both the rate and extent of tacrolimus absorption when administered to 15 healthy volunteers. The effect was most pronounced with a high-fat meal (848 kcal, 46% fat): mean AUC and Cmax were decreased 37% and 77%, respectively; Tmax was lengthened 5-fold. A high-carbohydrate meal (668 kcal, 85% carbohydrate) decreased mean AUC and mean Cmax by 28% and 65%, respectively. In healthy volunteers (N=16), the time of the meal also affected tacrolimus bioavailability. When given immediately following the meal, mean Cmax was reduced 71%, and mean AUC was reduced 39%, relative to the fasted condition. When administered 1.5 hours following the meal, mean Cmax was reduced 63%, and mean AUC was reduced 39%, relative to the fasted condition. In 11 liver transplant patients, Prograf administered 15 minutes after a high fat (400 kcal, 34% fat) breakfast, resulted in decreased AUC (27±18%) and Cmax (50±19%), as compared to a fasted state. Tacrolimus capsules should be taken consistently every day either with or without food because the presence and composition of food decreases its bioavailability.
- Distribution:
- Plasma protein binding is = approximately 99% and is independent of concentration over a range of 5-50 ng/mL.
- Tacrolimus is bound mainly to albumin and alpha-1-acid glycoprotein, and has a high level of association with erythrocytes. The distribution of tacrolimus between whole blood and plasma depends on several factors, such as hematocrit, temperature at the time of plasma separation, drug concentration, and plasma protein concentration. In a U.S. trial, the ratio of whole blood concentration to plasma concentration averaged 35 (range 12 to 67).
- Metabolism:
- Tacrolimus is extensively metabolized by the mixed-function oxidase system, primarily the cytochrome P-450 system (CYP3A).
- A metabolic pathway leading to the formation of 8 possible metabolites has been proposed. Demethylation and hydroxylation were identified as the primary mechanisms of biotransformation in vitro. The major metabolite identified in incubations with human liver microsomes is 13-demethyl tacrolimus. In in vitro studies, a 31-demethyl metabolite has been reported to have the same activity as tacrolimus.
- Excretion:
- The mean clearance following IV administration of tacrolimus is 0.040, 0.083, and 0.053, and 0.051 L/hr/kg in healthy volunteers, adult kidney transplant patients, adult liver transplant patients, and adult heart transplant patients, respectively.
- In man, less than 1% of the dose administered is excreted unchanged in urine. In a mass balance study of IV administered radiolabeled tacrolimus to 6 healthy volunteers, the mean recovery of radiolabel was 77.8±12.7%. Fecal elimination accounted for 92.4±1.0% and the elimination half-life based on radioactivity was 48.1±15.9 hours whereas it was 43.5±11.6 hours based on tacrolimus concentrations. The mean clearance of radiolabel was 0.029±0.015 L/hr/kg and clearance of tacrolimus was 0.029±0.009 L/hr/kg. When administered PO, the mean recovery of the radiolabel was 94.9±30.7%. Fecal elimination accounted for 92.6±30.7%, urinary elimination accounted for 2.3±1.1% and the elimination half-life based on radioactivity was 31.9±10.5 hours whereas it was 48.4±12.3 hours based on tacrolimus concentrations. The mean clearance of radiolabel was 0.226±0.116 L/hr/kg and clearance of tacrolimus 0.172±0.088 L/hr/kg.
- Specific Populations:
- Pediatric: Pharmacokinetics of tacrolimus have been studied in liver transplantation patients, 0.7 to 13.2 years of age. Following IV administration of a 0.037 mg/kg/day dose to 12 pediatric patients, mean terminal half-life, volume of distribution and clearance were 11.5±3.8 hours, 2.6±2.1 L/kg and 0.138±0.071 L/hr/kg, respectively. Following oral administration to 9 patients, mean AUC and Cmax were 337±167 ng·hr/mL and 48.4±27.9 ng/mL, respectively. The absolute bioavailability was 31±24%. Whole blood trough concentrations from 31 patients less than 12 years old showed that pediatric patients needed higher doses than adults to achieve similar tacrolimus trough concentrations. Pharmacokinetics of tacrolimus have also been studied in kidney transplantation patients, 8.2±2.4 years of age. Following IV infusion of a 0.06 (range 0.06 - 0.09) mg/kg/day to 12 pediatric patients (8 male and 4 female), mean terminal half-life and clearance were 10.2±5.0 (range 3.4-25) hours and 0.12±0.04 (range 0.06-0.17) L/hr/kg, respectively. Following oral administration to the same patients, mean AUC and Cmax were 181±65 (range 81-300) ng·hr/mL and 30±11 (range 14-49) ng/mL, respectively. The absolute bioavailability was 19±14 (range 5.2-56) %.
- Renal Impairment: Tacrolimus pharmacokinetics following a single IV administration were determined in 12 patients (7 not on dialysis and 5 on dialysis, serum creatinine of 3.9±1.6 and 12.0±2.4 mg/dL, respectively) prior to their kidney transplant. The pharmacokinetic parameters obtained were similar for both groups. The mean clearance of tacrolimus in patients with renal dysfunction was similar to that in normal volunteers.
- Hepatic Impairment: Tacrolimus pharmacokinetics have been determined in six patients with mild hepatic dysfunction (mean Pugh score: 6.2) following single IV and oral administrations. The mean clearance of tacrolimus in patients with mild hepatic dysfunction was not substantially different from that in normal volunteers. Tacrolimus pharmacokinetics were studied in 6 patients with severe hepatic dysfunction (mean Pugh score: >10). The mean clearance was substantially lower in patients with severe hepatic dysfunction, irrespective of the route of administration.
- Race: The pharmacokinetics of tacrolimus have been studied following single IV and oral administration of tacrolimus to 10 African-American, 12 Latino-American, and 12 Caucasian healthy volunteers. There were no significant pharmacokinetic differences among the three ethnic groups following a 4-hour IV infusion of 0.015 mg/kg. However, after single oral administration of 5 mg, mean (±SD) tacrolimus Cmax in African-Americans (23.6±12.1 ng/mL) was significantly lower than in Caucasians (40.2±12.6 ng/mL) and the Latino-Americans (36.2±15.8 ng/mL) (p<0.01). Mean AUC0-inf tended to be lower in African-Americans (203±115 ng·hr/mL) than Caucasians (344±186 ng·hr/mL) and Latino- Americans (274±150 ng·hr/mL). The mean (±SD) absolute oral bioavailability (F) in African-Americans (12±4.5%) and Latino-Americans (14±7.4%) was significantly lower than in Caucasians (19±5.8%, p=0.011). There was no significant difference in mean terminal T1/2 among the three ethnic groups (range from approximately 25 to 30 hours). A retrospective comparison of African-American and Caucasian kidney transplant patients indicated that African-American patients required higher tacrolimus doses to attain similar trough concentrations.
- Gender: A formal trial to evaluate the effect of gender on tacrolimus pharmacokinetics has not been conducted, however, there was no difference in dosing by gender in the kidney transplant trial. A retrospective comparison of pharmacokinetics in healthy volunteers, and in kidney, liver and heart transplant patients indicated no gender-based differences.
- Drug Interactions:
- Frequent monitoring of whole blood concentrations and appropriate dosage adjustments of tacrolimus are recommended when concomitant use of the following drugs with tacrolimus is initiated or discontinued.
- Telaprevir: In a single dose study in 9 healthy volunteers, coadministration of tacrolimus (0.5 mg single dose) with telaprevir (750 mg three times daily for 13 days) increased the tacrolimus dose-normalized Cmax by 9.3-fold and AUC by 70-fold compared to tacrolimus alone.
- Boceprevir: In a single dose study in 12 subjects, coadministration of tacrolimus (0.5 mg single dose) with boceprevir (800 mg three times daily for 11 days) increased tacrolimus Cmax by 9.9-fold and AUC by 17-fold compared to tacrolimus alone.
- Nelfinavir: Based on a clinical study of 5 liver transplant recipients, co-administration of tacrolimus with nelfinavir increased blood concentrations of tacrolimus significantly and, as a result, a reduction in the tacrolimus dose by an average of 16-fold was needed to maintain mean trough tacrolimus blood concentrations of 9.7 ng/mL. It is recommended to avoid concomitant use of Prograf and nelfinavir unless the benefits outweigh the risks.
- Rifampin: In a study of 6 normal volunteers, a significant decrease in tacrolimus oral bioavailability (14±6% vs. 7±3%) was observed with concomitant rifampin administration (600 mg). In addition, there was a significant increase in tacrolimus clearance (0.036±0.008 L/hr/kg vs. 0.053±0.010 L/hr/kg) with concomitant rifampin administration.
- Magnesium-aluminum-hydroxide: In a single-dose crossover study in healthy volunteers, co-administration of tacrolimus and magnesium-aluminum-hydroxide resulted in a 21% increase in the mean tacrolimus AUC and a 10% decrease in the mean tacrolimus Cmax relative to tacrolimus administration alone.
- Ketoconazole: In a study of 6 normal volunteers, a significant increase in tacrolimus oral bioavailability (14±5% vs. 30±8%) was observed with concomitant ketoconazole administration (200 mg). The apparent oral clearance of tacrolimus during ketoconazole administration was significantly decreased compared to tacrolimus alone (0.430±0.129 L/hr/kg vs. 0.148±0.043 L/hr/kg). Overall, IV clearance of tacrolimus was not significantly changed by ketoconazole co- administration, although it was highly variable between patients.
- Voriconazole (see complete prescribing information for VFEND): Repeat oral dose administration of voriconazole(400 mg every 12 hours for one day, then 200 mg every 12 hours for 6 days) increased tacrolimus (0.1 mg/kg single dose) Cmax and AUCτ in healthy subjects by an average of 2-fold (90% CI: 1.9, 2.5) and 3-fold (90% CI: 2.7, 3.8), respectively.
- Posaconazole (see complete prescribing information for Noxafil): Repeat oral administration of posaconazole (400 mg twice daily for 7 days) increased tacrolimus (0.05 mg/kg single dose) Cmax and AUC in healthy subjects by an average of 2-fold (90% CI: 2.01, 2.42) and 4.5-fold (90% CI 4.03, 5.19), respectively.
- Caspofungin (see complete prescribing information for CANCIDAS): Caspofungin reduced the blood AUC0-12 of tacrolimus by approximately 20%, peak blood concentration (Cmax) by 16%, and 12-hour blood concentration (C12hr) by 26% in healthy adult subjects when tacrolimus (2 doses of 0.1 mg/kg 12 hours apart) was administered on the 10th day of CANCIDAS 70 mg daily, as compared to results from a control period in which tacrolimus was administered alone.
- Advise patients to take the drug at the same 12-hour intervals everyday to achieve consistent blood concentrations
- Advise patients to take the drug consistently either with or without food because the presence and composition of food decreases the bioavailability of it
- Advise patients not to eat grapefruit or drink grapefruit juice during therapy
- Inform patients they are at increased risk of developing lymphomas and other malignancies, particularly of the skin, due to immunosuppression. Advise patients to limit exposure to sunlight and ultraviolet light by wearing protective clothing and use a sunscreen with a high protection factor
- Inform patients they are at increased risk of developing a variety of infections, including opportunistic infections, due to immunosuppression, and to contact their physician if they develop any symptoms of infection
- Inform patients that tacrolimus can have toxic effects on the kidney that should be monitored. Advise patients to attend all visits and complete all blood tests ordered by their medical team
- Inform patients that they are at risks of developing adverse neurologic effects including seizure, altered mental status, and tremor. Advise patients to contact their physician should they develop vision changes, deliriums, or tremors
- Inform patients that tacrolimus can cause hyperkalemia. Monitoring of potassium levels may be necessary, especially with concomitant use of other drugs known to cause hyperkalemia
- Inform patients that tacrolimus can cause high blood pressure which may require treatment with anti-hypertensive therapy
- Instruct patients to tell their healthcare providers when they start to stop taking all the medicines, including prescription medicines and non-prescription medicines, natural or herbal remedies, nutritional supplements and vitamins
- Instruct patients to tell their healthcare provider if they plan to become pregnant or breast-feed their infant
- Inform patients that tacrolimus can interfere with the usual responses to immunizations and theat they should avoid live vaccines
Indications
Dosing (Adult)
General Dosing & Administration Notes:
Dosing (Pediatric)
Renal Dosing
Warnings
Adverse Reactions
Overdose
Drug Interactions
Special Populations
Breasfeeding
Chemical Structure
Mechanism of Action
Pharmacokinetics
Counseling Points
MESH Terms & Keywords
|
|---|
|

