The
use of azathioprine (Imuran; Azasan) is common for a number of conditions, some
of which include the prevention of rejection of organ transplantation,
inflammatory bowel disease (in particular Crohn's disease), rheumatoid
arthritis, systemic lupus erythematosus, and several other immune mediated
chronic diseases.1-10 While not intuitively related to azathioprine,
allopurinol (Zyloprim, Aloprim) is used mainly for the prevention of gout
attacks, treatment of tophi, treatment of calcium oxalate calculi, and elevated
levels of uric acid in patients with leukemia, lymphoma and malignancies.11
In addition to these indications, the use of allopurinol along with
azathioprine based therapy has been done to improve outcomes in pediatric and
adult patients with inflammatory bowel disease, prevention of rejection in
organ transplantation, and reducing thiopurine-induced hepatotoxicity.3-8 Regardless of the
reason for coadministration of allopurinol and azathioprine, their concomitant
use should be done with caution and close monitoring or, only be a clinician with expertise in managing such patients.
What happens if allopurinol and azathioprine are used together?
The
simple answer is that the patient incurs significant risk for potentially life
threatening reductions in white blood cells (WBC). This is especially
true if the dose of azathioprine is not reduced. A significant drug-drug
interaction occurs that shunts azathioprine's primary route of metabolism to a
pathway that favors the production of active metabolites that are also known to
cause side effects. These active metabolites are known to decrease
replication and activation of WBCs while also inducing apoptosis (programmed
cell death) of WBCs (see figure).1,3,4,8,11-16 The cumulative effect of
this interaction can be a dramatic reduction in WBCs.
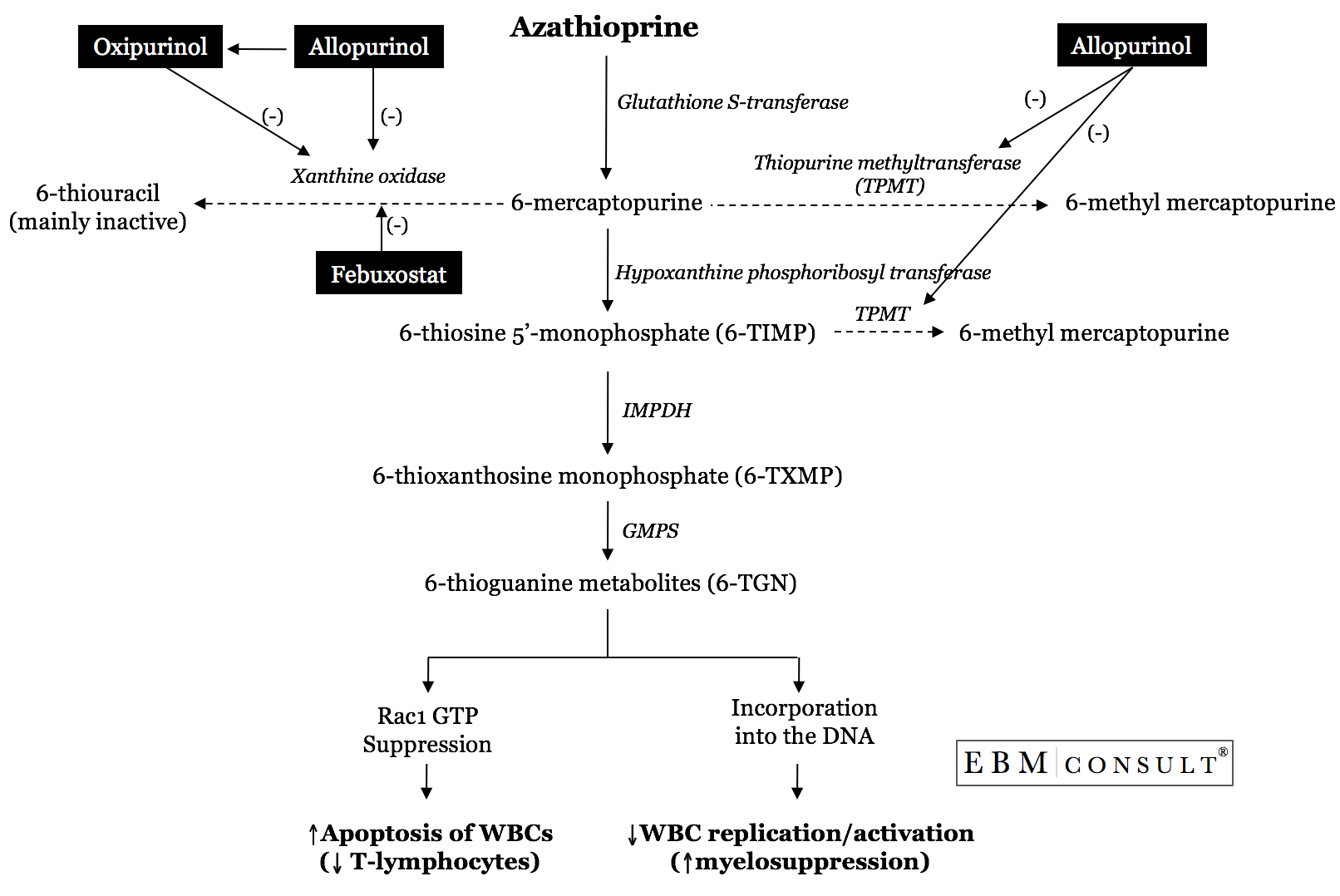
What is the
mechanism of this drug-drug interaction and the resulting reduction in WBCs?
After
oral administration and absorption, approximately 90% of azathioprine is
converted to 6-mercaptopurine (6-MP).1,3,4,15 At this point, 6-MP can
undergo metabolism by 1 of 3 different pathways; 2 of these pathways serve to
inactivate 6-MP and produce metabolites that are largely inactive while the
third pathway results in the production of active metabolites. The 2
inactivating pathways are carried out separately by the enzymes thiopurine
methyltransferase (TPMT) and xanthine oxidase (XO). The inactivation
pathway catalyzed by TMPT is straight forward, but unfortunately is subject to
the influence of genetic polymorphism.12 The inactivating XO pathway is also
straight forward in terms of converting 6-MP to the inactive metabolite
6-thiouracil. However, anything that inhibits XO will shunt 6-MP down the
pathway that results in the formation of the active 6-thioguanine (TGN) and
6-methyl-MP (MMP) metabolites. This is where allopurinol interacts with
azathioprine.
Allopurinol
and its metabolite (oxipurinol) are both known inhibitors of xanthine oxidase.11
Normally, XO is needed to convert the purine base hypoxanthine to xanthine and
xanthine to then uric acid. Allopurinol is a structural analogue of
hypoxanthine and binds to XO, thereby inhibiting its effect on purine
metabolism. Allopurinol may also have effects on TPMT activity as one study showed a reduction in methylated metabolites with the combination.16 Therefore, the coadministration of allopurinol with
azathioprine results in an increased shunting of 6-MP down the pathway that are then incorporated into DNA
resulting in decreased WBC replication/activation and/or suppression of
ras-related C3 botulinum toxin substrate 1 (Rac1) which can facilitate
apoptosis (programmed cell death) of WBC's.14
The
Details for Those Who Want Them:
- Normally,
CD28 costimulation of CD4+ T cells results in activation of Vav guanine
nucleotide exchange factors (GEF), thereby resulting in the conversion of
Rac1-bound GDP to Rac1-bound GTP.14 This, in turn, results in the
activation of MEKK/IkB/NF-kB pathway and STAT-3 activation that ultimately
enhances the amount of bcl-xL levels that provide an important
"antiapoptotic" signal. Therefore, anything that reduces the
expression or activation of bcl-xL would increase the mitochondrial
pathway of apoptosis. This is one of the mechanisms for
azathioprine (specifically the 6-TGN triphosphate metabolite) induced
inhibition of WBCs.14 6-TGN triphosphate blocks the activation of
Rac1-bound GDP to Rac1-bound GTP that is needed to activate the "antiapoptotic"
pathway mentioned above.
In
most studies where allopurinol was purposefully added to azathioprine regimens
for improved efficacy, the dose of azathioprine was decreased to avoid
life-threatening reductions in WBC.3,4,8 Despite this, some patients
still experienced significant drops in neutrophil counts to <1,000 cells/mm3.4
This appears to be reversible with the discontinuation of therapy.4
Failing to recognize or monitor for this interaction, whether being done for
synergy or for treatment of separate conditions, can increase the risk for
life-threatening pancytopenia or myelotoxicity. While no definitive data
are available, it is assumed that febuxostat (Uloric) would cause the same drug
interaction as allopurinol since it is also an inhibitor of XO.
References:
- Azathioprine tablets (Azasan) product package insert. Salix Pharmaceuticals, Inc., Wilmington, NC. 2003.
- McGeown
MG, Douglas JF, Donaldson RA et al. Ten-year results of renal
transplantation with azathioprine and prednisolone as only
immunosuppression. Lancet 1988;1:983-5.
- Sparrow
MP, Hande SA, Friedman S et al. Effect of allopurinol on clinical
outcomes in inflammatory bowel disease nonresponders to azathioprine or
6-mercaptopurine. Clin Gastroenterol Hepatol 2007;5:209-14.
- Rahhal
RM, Bishop WP. Initial clinical experience with allopurinol-thiopurine
combination therapy in pediatric inflammatory bowel disease. Inflamm
Bowel Dis 2008;14:1678-82.
- Chocair
P, Duley J, Simmonds HA et al. Low-dose allopurinol plus
azathioprine/cyclosporine/prednisolone, a novel immunosuppressive
regimen. Lancet 1993;342:83-4.
- A,
Evans C, Keevil B et al. Effect of allopurinol on the metabolism of
azathioprine in heart transplant patients. Transplant Proc
1998;30:1127-9.
- Chocair
PR, Duley JA, Cameron JS et al. Does low-dose allopurinol, with
azathioprine, cyclosporine and prednisolone, improve renal transplant
immunosuppression? Adv Exp Med Biol 1994;370:205-8.
- Ansari
A, Elliott T, Baburajan B et al. Long-term outcome of using
allopurinol co-therapy as a strategy for overcoming thiopurine
hepatotoxicity in treating inflammatory bowel disease. Aliment
Pharmacol Ther 2008;28:734-41.
- De Silva M, Hazleman BL. Long-term azathioprine in rheumatoid arthritis: a double-blind study. Ann Rheum Dis 1981;40:560-3.
- Ginzler
E, Sharon E, Diamond H et al. Long-term maintenance therapy with
azathioprine in systemic lupus erythematosus. Arthritis Rheum
1975;18:27-34.
- Allopurinol tablets product package insert. Watson Laboratories, inc.; Corona, CA. January 2006.
- Dubinsky
MC, Lamothe S, Yang HY et al. Pharmacogenomics and metabolite
measurement for 6-mercaptopurine therapy in inflammatory bowel disease.
Gastroenterology 2000;118:705-13.
- Lepage GA. Basic biochemical effects and mechanism of action of 6-thioguanine. Cancer Res 1963;23:1202-6.
- Tiede
I, Fritz G, Strand S et al. CD28-dependent Rac1 activation is the
molecular target of azathioprine in primary human CD4+ T lymphocytes. J
Clin Invest 2003;111:1133-45.
- Lennard L. The clinical pharmacology of 6-mercaptopurine. Eur J Clin Pharmacol 1992;43:329-39.
- Smith MA, Blaker P, Marinaki AM et al. Optimising outcome on thiopurines in inflammatory bowel disease by co-prescription of allopurinol. J Crohns Colitis 2012;6(9):905-12.


